Protective Effects of Capparis zeylanica Linn. Leaf Extract on Gastric Lesions in Experimental Animals
-
 R.Sini, Karanayil
Karanayil R Sini, M. pharm., Department of Pharma-ceutical Chemistry, Grace College of Pharmacy, Palakkad, India , E-mail: sinikaranayil@gmail.com
R.Sini, Karanayil
Karanayil R Sini, M. pharm., Department of Pharma-ceutical Chemistry, Grace College of Pharmacy, Palakkad, India , E-mail: sinikaranayil@gmail.com
-
Department of Pharmaceutical Chemistry, Grace College of Pharmacy, Palakkad, India
-
N.Sinha, Barij
-
Department of Pharmaceutical Sciences, Jharkhand, India
Abstract: The aim of the study was to study the anti-ulcer activity of the methanolic extract of the leaves of Capparis zeylanica Linn on experimental animal models. The methanol extract of Capparis zeylanica Linn. leaves was investigated for anti-ulcer activity against aspirin plus pylorus ligation induced gastric ulcer in rats. HCl-Ethanol induced ulcer in mice and Indomethacin induced ulcer in rats at 200 mg/kg body weight p.o. A significant (p<0.01, p<0.001) anti-ulcer activity was observed in all the models. Pylorus ligation showed significant (p<0.01) reduction in gastric volume, free acidity and ulcer index as compared to control. It also showed 88.5% ulcer inhibition in HCl-ethanol induced ulcer and 83.78% inhibition in Indomethacin induced ulcer.
Introduction :
The exact pathogenesis of ulcer continues to elude scientists and medical researchers, but a common ground has been proposed. Ulcers are produced when any factor causes an imbalance between the protective factors (mucus and bicarbonate) and aggressive factors (acid and pepsin) in the stomach. The disease has been reported to have high recurrence and mortality rates especially in complicated cases (1,2). Such factors could range from natural causes (gastric cancer), infections (Helicobacter pylori-H. pylori), and lifestyle (drugs, non steroidal anti-inflammatory agents, alcohol, stress and cigarette smoking) (3,4). Current treatment of ulcers in developing countries has been largely suppression of pain, with little or no strategy aimed at a cure. Herbal medicine is fast emerging as an alternative treatment to available synthetic drugs for treatment of ulcer possibly due to lower costs, availability, fewer adverse effects and perceived effectiveness.
Gastric ulcer, one of the most widespread, is believed to be due to an imbalance between aggressive and protective factors (5). The gastric mucosa is continuously exposed to potentially injurious agents such as acid, pepsin, bile acids, food ingredients, bacterial products (H. pylori) and drugs (6). These agents have been implicated in the pathogenesis of gastric ulcer including enhanced gastric acid and pepsin secretion, inhibition of prostaglandin synthesis and cell proliferation growth, diminished gastric blood flow and gastric motility (7). The goals of treating peptic ulcer disease are to relieve pain, heal the ulcer and prevent ulcer recurrence. Currently there is no cost-effective treatment that meets all these goals. Hence, efforts are on to find a suitable treatment from natural product sources.
Many tropical herbs have been scientifically reported to possess potent anti-ulcer activity (8-10). Capparis zeylanica Linn. (C. zeylanica) commonly known as ‘Asadhua’ in Oriya and ‘Ardanda’ in Hindi is a branched thorny, sub-scandent climbing shrub found throughout India (11). It has been used as a ‘Rasayana’ drug in the traditional Ayurvedic system of medicine. In Northern India, the leaves are widely used as counter-irritant, febrifuge and as a cataplasm in swellings (12), boils and piles (13). Traditionally it is also used as antidote to snake bite, to cure swelling of testicle, small pox, boils, cholera, colic, hemiplagia, neuralgia, sores, pneumonic and pleurisy (14).
Whole plant showed the presence of saponin, p-hydroxybenzoic, syringic, vanillic, ferulic and p-coumanic acid. Leaves and seeds showed presence of β-carotene, thioglycoside, glycocapparin, n-tricortane, α-amyrin and fixed oil where as root bark showed presence of an alkaloid, a phytosterol, a water soluble acid and a mucilaginous substance (15,16). Capparis species has been reported to have anthelmintic, anti microbial and anti inflammatory activities (17,18). The dried leaves of few species from the genus Capparis, steeped in vinegar were reported to be used for treatment of ulcers (19).
Materials and Methods :
Plant collection
The fresh leaves of C. zeylanica Linn (Capparidaceae), collected at the flowering stage in the month of March 2010 from the tribal areas of Palakkad district, Kerala state, South India were authenticated by the Botanical survey of India, Combatore, Tamilnadu (BSI). A voucher specimen (no.BSI/SRC/5/23/10-11/Tech-565) is deposited in the departmental herbarium.
Preparation of extract
The leaves of C. zeylanica were shade dried and reduced to coarse powder in a mechanical grinder. The powdered material obtained was soxhlet extracted using methanol and the extract obtained was evaporated at 45°C to get a semisolid mass. The extract thus obtained was subjected to phytochemical analysis. The percentage yield of methanolic extract was found to be 36.50% w/w and was used for further studies (20).
Animals
Healthy adult albino rats of male sex weighing between 100-180 g obtained from Kerala Veterinary College, Mannuthy, Kerala. The animals were housed under standard conditions of light/dark at 12/12 hr cycle. They were fed with commercial pellet diet and water ad libitum. All animal experiments were carried out in accordance with guidelines of CPCSEA and the study was approved by the Institutional Animal Ethics Committee (NCP/IAEC/CLEAR/05/03/2007-08).
Acute oral toxicity studies
A safe oral dose of the extract was determined by acute oral toxic class method of Organization of Economic Co-operation and Development (OECD) as per 423 guidelines (21).
Anti-ulcer activity
Three models (ethanol, aspirin, and indomethacin) with effective induction of ulcer experimentally in rats were employed to evaluate the anti-ulcer activity of the methanolic extract of C. zeylanica. All the rats used were fasted for 18 hr hours but were given water ad libitum till the start of the experiment.
Ethanol-induced ulcer
Thirty fasted animals were used in three groups of six animals each. Groups A and B received 5 ml/kg (p.o) of 3% Tween 80 (negative control) and 100 mg/kg p.o sucralfate (Antepsin®) while rats in group C were given 200 mg/kg of MECZ orally (p.o), respectively. After 1 hr all animals received 1 ml/kg of 80% ethanol (Sigma-Aldrich, Germany) orally. The rats were sacrificed with chloroform (Sigma-Aldrich, Germany) anaesthesia after 1 hr. The stomachs were isolated, washed gently under clean flowing water and cut open along the greater curvature. The stomachs were then fixed in 10% formalin and craters observed and ulcer scores were recorded (10).
Aspirin-induced ulcer
The wistar albino rats weighing 100-200 g of either sex were divided into three groups, each group consists of 6 animals. All the animals received 200 mg/kg of aspirin once daily for three days. Group1 served as control received 5 ml/kg (p.o) of 3% Tween 80, group 2 treated with 50 mg/kg (p.o) ranitidine as standard and group 3 treated with 200 mg/kg (p.o) methanol extract of C. zeylanica. On the fourth day pylorus part was ligated following 36 hr fasting (22). Four hrs after the pylorus ligation the animals were sacrificed by decapitation. The stomach was opened and the ulcer index was determined (23). The gastric contents were titrated against 0.01 N NaOH to determine the free acidity and total acidity (24).
Indomethacin-induced ulcer
Animals (three groups of six rats each) in groups received 5 ml/kg (p.o) of 3% Tween 80, Omeprazole 20 mg/kg (p.o) and 200 mg/kg (p.o) of extract, respectively. After 30 min, indomethacin 40 mg/kg (p.o) was administered to each rat. After 8 hr of drug treatment, stomachs were isolated, cut and ulcers counted as before (25).
Statistical analysis
Ulcer indices were shown as the mean standard error of mean and level of ulcer protection presented as percentage inhibition. The statistical analysis was carried out using one-way ANOVA followed by Dunnetts multiple comparisons using graph pad in stat3.
Result :
Preliminary phytochemical screening of the Methanolic Extract of C. zeylanica (MECZ) revealed that presence of alkaloids, flavonoids, carbohydrates, glycosides, tannins, terpenoids, phenols and absence of fixed oils and steroids. No visible change was observed in any animal and all survived till 24 hr; thus based on preliminary study, different extracts of C. zeylanica were found to be safe for further biological studies, as no lethality was observed at 1000 mg/kg (p.o) in mice. Acute toxicity results showed that the LD50 was greater than 5000 mg/kg.
In aspirin plus pylorus ligation induced gastric ulcer, the methanol extract of C. zeylanica showed significant reduction in gastric volume, free acidity and ulcer score (% ulcer inhibition 57.83% as compared to control (Table 1). From table 2 it can be observed that the number of lesions in HCl- ethanol induced peptic ulcer group was significantly high and the methanol extract of C. zeylanica pre-treated group depicted marked reduction (p<0.01) in gastric lesion (% ulcer inhibition 88.5% ) as compared to control (Table 2).
Results for indomethacin model can be viewed in table 3. There was an absolute production of severe ulcers in all the rats using this model and ulcer inhibition was seen in all the treatment groups. Ulcer inhibition was highest with 200 mg/kg MECZ (83.78 %, p>0.01) and lowest with Sucralfate (48.74%).
Discussion :
The anti-ulcer activity of the methanol extract of C. zeylanica against ethanol, aspirin and indomethacin-induced ulcers was established in this study. Results of acute toxicity showed that the plant is safe as exemplified by its use as food in domestic and wild animals. The extract protected the stomach against ethanol’s necrotic damage and its effect was more pronounced than sucralfate, a cytoprotective agent.
Ethanol has been shown to increase the risk of ulcer in humans (2), but produces potent ulceration in rats (26). Ethanol challenge induces gastric injury due to production of oxygen free radicals leading to increased lipid peroxidation, which causes damage to cell and cell membrane presenting as red streaks of sores (27). The protection by the extract of this type may suggest a possible cytoprotective mechanism of action.
Indomethacin model produces its ulceration by mechanism well understood. It causes an inhibition of the production of endogenous cytoprotective prostaglandins (28). However, with the inhibition of ulcer in this model by the extract, it is possible that the plant produces a cytoprotection similar to those of Omeprazole in the indomethacin model. The secondary metabolites identified may also have been responsible for the anti-ulcer activity of this plant as flavonoids have been reported to possess anti-ulcer activity in various experimental models of ulcers (29).
In aspirin plus pylorus ligation induced gastric ulcer model, the methanol extract of C. zeylanica reduced the gastric volume, free acidity and ulcer index thus showing the antisecretory mechanism involved in the extracts for their anti-ulcerogenic activity. Ulcer index parameter was used for the evaluation of anti-ulcer activity since ulcer formation is directly related to factors such as gastric volume, free and total acidity. In case of vehicle control, aspirin plus pylorus ligation increased the acid secretion, which in turn caused increase in gastric volume, low pH, increased free and total acidity resulting into increase in ulcer index (30).
Ulcer protection may be attributed to any of these phytochemical constituents as flavonoids, tannins and saponins which have been shown to produce anti-ulcerogenic and anti-gastric activity (31). However, until specific constituents are isolated and characterized, exact mechanism of action cannot be ascertained. Studies on the sub-acute and chronic toxicity of the extract are however in progress.
Acknowledgement :
The authors are grateful to staff of forest department, Attapady for their kind help during field visits and tribal people who shared their traditional knowledge regarding medicinal plants during our field visits.
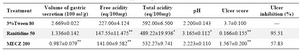
Table 1. Effect of methanol extract of C. zeylanica (MECZ) on gastric secretion, acidity, pH and ulcer score in aspirin plus
pylorus ligated rats
n= 6, Values are expressed as mean ± SEM, p<0.05 when compared with control
*p<0.05, Significant as compared with control group; **p<0.01 when compared with control
|
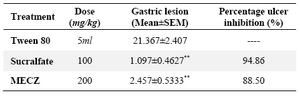
Table 2. Effect of methanolic leaf extract of C. zeylanica (MECZ) on ethanol induced ulcer in rats
n= 6, Values are expressed as mean ± SEM, p<0.01 when compared with control
*p<0.05, Significant as compared with control group; **p<0.001 when compared with control
|
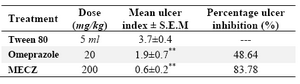
Table 3. Effect of methanol extract of C. zeylanica (MECZ) on indomethacin induced ulcer in rats
n= 6, Values are expressed as mean±SEM, p<0.01 when compared with control
*P<0.05, Significant as compared with control group; **p<0.001 when compared with control
|
|