Conjugation of R-Phycoerythrin to a Polyclonal Antibody and F (ab')2 Fragment of a Polyclonal Antibody by Two Different Methods
-
Mahmoudian, Jafar
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Jeddi-Tehrani, Mahmood
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Immune and Gene Therapy Lab, Cancer Center Karolinska, Karolinska Institute, Stockholm, Sweden
-
Rabbani, Hodjattallah
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Immune and Gene Therapy Lab, Cancer Center Karolinska, Karolinska Institute, Stockholm , Sweden
-
Mahmoudi, Ahmad Reza
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Akhondi, Mohammad Mehdi
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Zarnani, Amir-Hassan
-
Nanobiotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Balaei Goli, Leila
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Babaei, Mahdokht
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
 Ghods, Roya
M.Sc., Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran, P.O. Box: 19615-1177, Tel: +982122432020 Fax: +0982122432021 E-mail: ghods@avicenna.ac.ir
Ghods, Roya
M.Sc., Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran, P.O. Box: 19615-1177, Tel: +982122432020 Fax: +0982122432021 E-mail: ghods@avicenna.ac.ir
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
Abstract: R-Phycoerythrin (R-PE), a fluorescent protein from phycobiliprotein family, is isolated from red algae. Conjugation of antibodies to R-PE facilitates multiple fluorescent staining methods. In the present study polyclonal antibodies and polyclonal F(ab')2 fragment antibodies were conjugated to R-PE by two different methods. The efficiency of the methods was evaluated using Immunocytochemistry (ICC) and Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE). In the first conjugation method, PE was attached to SMCC linker followed by conjugation of antibody to PE-SMCC. In the second method, SH groups were added onto R-PE molecule, while the antibody was attached to SPDP linker. Then, the antibody-SPDP molecule was conjugated to R-PE. Our results showed that the two conjugation methods did not have any abrogative effects on the antibody binding activity.
Introduction :
R-Phycoerythrin (R-PE) is one of the most commonly-used fluorescent dyes for fluorescent staining and other immunoassays. R-PE is a large protein (approximately 240 kDa) containing 25 fluors (1). R-PE has three types of subunits: alpha (~20,000 daltons), beta (~20,000 daltons) and gamma (~30,000 daltons) and its subunit structure [(alpha-beta) 6 gamma] has been determined (2). Typically, only one PE molecule is conjugated to one antibody molecule (3).
Nonetheless, by virtue of its huge absorption coefficient and almost perfect quantum efficiency it is one of the brightest dyes used today. It emits at about 570 nm, and is excited by an argon laser tuned to 488 nm; however, much more sensitive detection is obtained with a laser tuned to 533 nm (3).
In this study we conjugated R-PE to antibodies by two various methods. Firstly, R-PE was attached to SMCC linker; and then, the antibody was attached to PE-SMCC (3). Secondly, SH groups were added onto PE molecule, while the antibody was attached to SPDP linker. Then, the antibody-SPDP molecules were conjugated to R-PE (4 - 6).
Materials and Methods :
Antibodies (Abs)
For the purpose of conjugation, three different antibodies were prepared at Avicenna Research Institute, Tehran, Iran.
These antibodies included sheep anti mouse immunoglobulins adsorbed against human immunoglobulins [Sh?M Ig (human Ig ads)], F(ab')2 fragment of sheep anti mouse immunoglobulins adsorbed against human immunoglobulins [Sh?M Ig F(ab')2 (human Ig ads)], sheep anti human immunoglobulins adsorbed against mouse immunoglobulins [Sh?H (Mouse Ig Ads)].
Conjugation of R- PE to a reduced antibody by SMCC linker:
Sulfosuccinimidyl- 4-(N-maleimidomethyl) cyclohexane-1-carboxylate (Sulfo-SMCC) (Sigma- Aldrich, Wisconsin, USA) is a water-soluble heterobifunctional linker. It contains an amine-reactive N-hydroxysuccinimide (NHS ester) and a sulfhydryl-reactive maleimide group. First, 200 µg sulfo-SMCC was attached to 1 mg R-PE (2-3 mg/ml) by the amine-reactive end for 1 hr with shaking in Phosphate Buffered Saline (PBS); then, the excess sulfo-SMCC was removed by dialysis in PBS at 4 ?C overnight. The antibody was reduced by 20 mM Dithiothreitol (DTT) for 30 min without shaking; and then, DTT was rapidly removed by dialysis in PBS. The reduced antibody was immediately added to PE-SMCC where the sulfhydryl-reactive maleimide end in PE-SMCC was used to attach PE- SMCC to the reduced antibody. Then, the mixture was mixed for 6 hr at Room Temperature (RT). Finally, free SH groups on the antibody molecules were blocked by 40 mM N- Ethylmaleimide (NEM) for 30 min at RT (3).
Conjugation of thiolated PE to the antibody by SPDP linker
R-PE was thiolated by Traut's Reagent (2-Iminothiolane or 2-IT) (Sigma-Aldrich) for 1.5 hr at RT, while the antibody was attached to a heterobifunctional linker called SPDP (N -succinimidyl 3-(2-pyridyldithio)-propionate) (Uptima, Montiucon, Cedex, France) in PBS. Finally, thiolated R-PE was mixed with Ab-SPDP; resulting in Ab-SPDP-PE.
Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE)
To analyze conjugate quality, equal amounts of protein (10 µg) were run on 12.5% SDS-PAGE at 100 V for 2 hrs under discontinuous non-reducing condition using a Mini-Protean III electrophoresis apparatus (Bio-Rad, Hercules, CA).
Cell culture
A mouse IgG-producing hybridoma cell line was grown in RPMI 1640 medium containing 10% (v/v) fetal calf serum (Invitrogen, California, USA) and 1% penicillin/ streptomycin (Sigma- Aldrich) at 37 °C in the presence of 5% Co2.
Immunocytochemistry
Two different cell types were used in this experiment. A mouse IgG-producing hybridoma cell line and human B lymphocytes as part of Peripheral Blood Mononuclear Cells (PBMC) that were prepared from whole blood by Ficoll separation (7). Forty thousand cells were added onto glass slides. After drying for 2 hr at RT, they were fixed by 2% formaldehyde. The fixed cells were washed and then blocked by 5% sheep serum. R-PE conjugated antibodies were added. Cells were washed with PBS and then observed directly under a fluorescent microscope (Olympus, Tokyo, Japan).
Result :
Electrophoretic pattern of both PE conjugated whole antibody and F(ab')2 fragments by SMCC linker
Figures 1 and 2 show electrophoretic mobility patterns of R-PE conjugated whole Sh?M Ig and F(ab')2 fragment of Sh?M Ig by SMCC linker in SDS-PAGE, respectively. Considering the fact that the conjugation of antibodies to R-PE may involve one or more R-PE subunits for each antibody molecule, the conjugation materials may show different electrophoretic mobility properties as reflected by a smear rather that a single sharp band in SDS-PAGE (2).
Electrophoretic pattern of both PE conjugated whole antibodies and F(ab')2 fragments by SPDP linker
Figure 3 shows electrophoretic mobility patterns of R-PE conjugated whole and F(ab')2 fragments of Sh?M Ig by SPDP in SDS-PAGE. Similar to SMCC linker method, the conjugation method using SPDP linker also resulted in smears because of different numbers of R-PE subunits bound per Ab molecule.
Immunocytochemistry analysis of antibodies conjugated by SMCC linker
To analyze the specific binding of R-PE conjugated antibodies by SMCC linker method, conjugated whole Sh?M Ig and its F(ab')2 fragments were studied by ICC on a mouse IgG-producing hybridoma cell line. The R-PE conjugated Sh?H Ig as the control antibody was tested for its specific reactivity on human B lymphocyte in ICC assay as depicted in figures 4, A and B. Both Sh?M Ig – R-PE and its R-PE conjugated F(ab')2 fragments could successfully bind to the cells (Figures 5A and B) while the negative control, R-PE conjugated Sh?H Ig (Figure 5C) did not bind to these cells.
Immunocytochemistry analysis of antibodies conjugated by SPDP linker
Specific binding of whole R-PE conjugated Sh?M Ig and its F(ab')2 fragments by SPDP linker was tested by ICC on the mouse IgG-producing hybridoma cell line. As shown in figure 6 both antibody conjugates reacted to the cells (Figures 6A and B), while its negative control (Sh?H Ig-R-PE) was negative as shown in figure 6 C.
Discussion :
Though there are different methods to conjugate R-PE to antibodies, optimization of an efficient method is crucial. Conjugating R-PE molecules to antibodies without other adverse byproducts is our main goal in this conjugation. Considering the fact that R-PE – R-PE and Ab – Ab conjugates are not useful, a proper conjugation should attach R-PE to antibody specifically. The commonly used homobifunctional linkers, like glutaraldehyde, have some disadvantages. They result in high
levels of adverse byproducts such as Ab – Ab and R-PE – R-PE conjugates; in addition, their chemical process is hard to control and may lead to unwanted polymerization of materials (8). Hence, in order to conjugate R-PE to Abs, the most efficient method is using heterobifunctional linkers (9). These linkers usually have two different end groups, an amine reactive group and a sulfhydryl reactive group, which provide a feasible tool to conjugate different molecules.
Selecting an appropriate method to attach two different molecules depends on their structure or surface active groups. On account of the fact that the surface of R-PE molecule contains many amine groups (10), and antibodies contain many intra-disulfide bonds (11), these different properties could be used as a significant tool during an appropriate bioconjugation. Therefore, in our first conjugation method a heterobifunctional linker, sulfo-SMCC, was used to link the amine groups on R-PE molecule to the sulfhydryl groups in the reduced antibody molecules.
However, this notion is challenged by some researchers who prefer to use the amine groups of antibody (12). To examine this possibility in our system, we used the second conjugation method in which SPDP linker was used to conjugate thiolated R-PE to amine groups on the intact antibody molecules. The results of experiments showed that the antibodies retained their binding ability in both systems.
Acknowledgement :
This work was supported by a grant from Avicenna Research Institute.
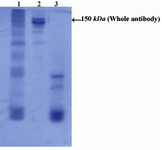
Figure 1. Electrophoretic pattern of R-PE conjugated ShM Ig by SMCC linker in non-reduced 12.5% SDS-PAGE: Lane 1 shows R-PE conjugated ShM Ig. Lane 2 shows unconjugated ShM Ig. Lane 3 shows R-PE alone
|
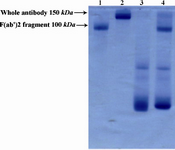
Figure 2. Electrophoretic pattern of R-PE conjugated F(ab')2 fragment of ShM Ig by SMCC linker in non-reduced 12.5% SDS-PAGE. Lane 1 shows F(ab')2 fragment of ShM Ig (100 kDa). Lane 2 shows IgG molecule (150 kDa) as protein marker. Lane 3 shows R-PE alone. Lane 4 shows R-PE conjugated F(ab')2 fragment of ShM Ig
|
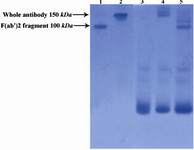
Figure 3. Electrophoretic pattern of R-PE conjugated ShM Ig and F(ab')2 fragment of ShM Ig by SPDP linker in non-reduced 12.5% SDS-PAGE. Lane1 shows unconjugated F(ab')2 fragment of ShM Ig. Lane 2 shows unconjugated ShM Ig. Lane 3 shows R-PE alone. Lane 4 shows R-PE conjugated ShM Ig. Lane 5 shows R-PE conjugated F(ab')2 fragment of ShM Ig
|
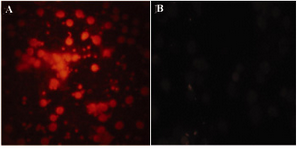
Figure 4. Immunocytochemistry analysis of R-PE conjugated ShH Ig on human B lymphocytes. A) ShH (Mouse Ig Ads)-PE (1 mg/ml dilution: 1/100). B) ShM Ig (Human Ig Ads)-PE (1 mg/ml dilution: 1/100)
|
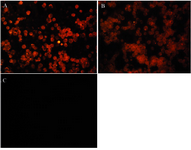
Figure 5. Immunocytochemistry analysis of ShM Ig – R-PE and its R-PE conjugated F(ab')2 fragments by SMCC linker on a mouse IgG-producing hybridoma cell line. A) ShM Ig (Human Ig Ads)-PE (1 mg/ml dilution: 1/100). B) F(ab')2 fragment of ShM Ig (Human Ig Ads)-PE (1 mg/ml dilution: 1/100). C) ShH (Mouse Ig Ads)-PE (1 mg/ml dilution: 1/100)
|
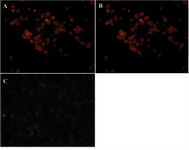
Figure 6. Immunocytochemistry analysis of ShM Ig – R-PE and its R-PE conjugated F(ab')2 fragments by SPDP linker on a mouse IgG-producing hybridoma cell line. A) ShM Ig (Human Ig Ads)-PE (1 mg/ml dilution: 1/100). B) F(ab')2 fragment of ShM Ig (Human Ig Ads)-PE (1 mg/ml dilution: 1/100). C) ShH (Mouse Ig Ads)-PE
(1 mg/ml dilution: 1/100)
|
|