Lack of Association Between Tumor Necrosis Factor-alpha -308 G/A Polymorphism and Risk of Developing Late-Onset Alzheimer's Disease in an Iranian Population
-
Manoochehri, Mehdi
-
Genetics Research Center, University of Social Welfare and Rehabilitation Sciences , Tehran, Iran
-
Kamali, Koorosh
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
-
Epidemiology and Biostatistics Department, Tehran University of Medical Sciences , Tehran, Iran
-
Rahgozar, Mehdi
-
Epidemiology and Biostatistics Department, University of Social Welfare and Rehabilitation Sciences , Tehran, Iran
-
Ohadi, Mina
-
Genetics Research Center, University of Social Welfare and Rehabilitation Sciences , Tehran, Iran
-
 Khorram Khorshid, Hamid Reza
M.D., Ph.D., Genetic Research Centre, University of Social Welfare and Rehabilitation Sciences, Tehran, Iran, Tel: +98 21 22180138, E-mail: hrkk1@uswr.ac.ir
Khorram Khorshid, Hamid Reza
M.D., Ph.D., Genetic Research Centre, University of Social Welfare and Rehabilitation Sciences, Tehran, Iran, Tel: +98 21 22180138, E-mail: hrkk1@uswr.ac.ir
-
Genetics Research Center, University of Social Welfare and Rehabilitation Sciences , Tehran, Iran
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR , Tehran, Iran
Abstract: Late-onset Alzheimer's Disease (LOAD) is a neurodegenerative disorder and the most common form of dementia affecting people over 65 years old. Alzheimer’s disease is a complex disease with multi-factorial etiology. Inflammation has been approved to have an important role in the pathogenesis of Alzheimer’s disease (AD). TNF-a is a main pro-inflammatory cytokine that plays an essential role in initiation and regulation of inflammatory responses. Several studies have shown the probable association of polymorphism at TNF-a gene’s promoter with AD pathogenesis. This study was performed to determine whether this polymorphism contributes to the risk for late-onset Alzheimer's disease (LOAD) in Iranian population. One hundred and forty AD patients and 158 healthy controls were recruited in the study. Following extraction of genomic DNA, using PCR/RFLP methods the genotype and allele frequencies were determined in case and control subjects. The statistical analysis showed no significant difference in the allele and genotype frequencies due to this polymorphism between the two groups. Also after stratifying the subjects by their APOE-e4 status, no significant association was observed. Our results suggest that Tumor necrosis factor-alpha (TNF-?)
-308 G/A is not a risk or protective factor for late-onset Alzheimer’s disease in Iranian population. However, to confirm these results further study with a bigger sample size may be required.
Introduction :
Late-onset Alzheimer's Disease (LOAD) is a neurodegenerative disorder and the most common form of dementia affecting people over 65 years old (1). Many previous studies suggest that inflammatory reactions in the central nervous system (CNS) play a funda-mental role in Alzheimer's disease pathogen-esis (2). The microglia are immune cells in the CNS that act as immunocompetent defense cells that initiate and regulate endogenous immune responses at the CNS (3).
The microglia has an important function in cellular responses to pathological lesions, such as Aß (amyloid ß) plaques (1). Exposures of these microglia to Aß plaques, activates them leading to an increase in expression of MHC class ??, some chemokines and proin-flammatory cytokines such as interleukin-1ß (IL-1ß), interleukin-6 and tumor necrosis factor alpha (TNF-a) (4). TNF-a is an import-ant proinflammatory cytokine that is upregu-lated in Alzheimer's disease patients (5-7).
After exposure to Aß plaques, the levels of TNF-a has been evaluated and shown to be increased in AD patients sera, CSF, cortex and glial cell cultures. It has been indicated that the excess levels of TNF-a kills human cortical neurons (8).
There are evidences that TNF-a, in com-bination with interferon-?, increase the production of Aß in vitro and inhibits the secretion of neuroprotective soluble amyloid Precursor Protein (APP). Also TNF-a is implicated in the up-regulation of cyclooxy-genase-2 (cox2) and increased amounts of damaging free radicals (7). A polymorphism in the promoter region of TNF-a-308 G/A has been implicated in promoter activity (9,10). The nucleotide variation in promoter of TNF-a gene, has been associated with AD patho-genesis. The A-allele has been reported to be associated with developing of LOAD in some population (11-13). These results indicated that TNF-a gene expression is afflicted in AD. Because the promoter regions are involved in regulation of gene expression and the results of mentioned studies indicated the association between -308 A-allele with AD, it is rational to determine the existence of any association between this polymorphism at the promoter region of TNF-a gene and the AD in Iranian population.
Materials and Methods :
To determine the possible role of -308G/A polymorphism in developing LOAD in our population, a case-control study was per-formed. A sample of 140 AD patients
(88 women, 52 men, mean age: 78.5±7.80 yr) and 158 healthy controls (105 women, 53 men, mean age: 77.14±6.95 yr) were recruited for the study. The diagnoses of Alzheimer’s disease in patients were confirmed by quali-fied psychiatrists according to the DSM-IV criteria and control subjects were selected through the assessment of their medical his-tories and physical conditions. The recruited subjects in control group did not have any known neuropsychiatric disorders with in the same age range. We assume genetic back ground (five major ethnic groups in Iranian population), occupation, education and gender as potential confounders (Table 1). After obtaining informed consent from participants or their families, the information regarding the age, sex, ethnicity, job and education were asked and recorded. Finally, 5 cc of peripheral blood samples were collected in tubes con-taining 200 µl of 0.5 M EDTA. Genomic DNA was extracted from peripheral blood using the salting-out method, and a pair of primers (Forward: 5'aggcaataggt tttgagggccat 3', Reverse: 5'tcctccctgctccgattcc g3') was used to amplify the relevant fragment and analyze the -308 G/A variation. The TNF-a-308 G/A genotyping was determined using PCR amplification and 8% polyacrylamid gel electrophoresis (PAGE). To amplify the 107 bp fragment, PCR was performed and the amplified products were digested using NCO1 restriction enzyme. Digestion by NCO1, produced 87 and 20 bp fragments for wild type (G allele) and intact 107 bp fragment for variant type (A allele).
The Logistic regression test and Chi square (?2) statistical analyses were performed to compare genotype and allele frequencies in the study groups. To assess the role of inter-action of APOE-e4 allele with this polymer-phism, the logistic regression was performed regarding the existing APOE-e4 PCR results with these samples. The different allele and genotype frequencies of APOE gene were determined in our previous study using PCR-RFLP method (14).
Result :
In total 298 DNA samples were analyzed using the PCR and Restriction Fragment Length Polymorphism (RFLP) methods. The samples consisted of 140 AD patients (88 women, 52 men, mean age: 78.5±7.80 yr) and 158 healthy controls (105 women, 53 men, mean age: 77.14±6.95 yr). The distributions of other covariates in the studied groups were similar (Table 1).
The distributions of TNF-a-308G/A geno-type and allele frequencies are summarized in table 2. We also categorized the participant’s APOE-e4 genotypes status based on the existing data from previous study (14). The statistical analysis of the patients’ genotype and allele frequencies showed that there was no statistically significant difference in TNF-a-308G/A genotype and allele frequencies between the AD patients and healthy subjects.
Next, we stratified the TNF-a-308G/A genotyping results according to their APOE-e4 status for synergic effects using logistic regression. After stratifying the APOE-e4 genotypes status there was no significant dif-ference between the AD patients and healthy controls, either (Table 3). Therefore, there was not statistically significant evidence for the APOE-e4 genotypes as a modifier for TNF-a-308 G/A polymorphism.
Discussion :
The inflammatory process has a fundamen-tal role in the pathogenesis of Alzheimer's disease. Recent studies indicate that inflam-mation is a powerful pathogenic factor in the disease process. Increased production of amyloid-beta peptide can activate the innate immunity system and evoke Alzheimer's pathology (15). Previous studies have indicated a decreased risk for developing Alzheimer's disease in Anti-inflammatory (AI) drug users (16). Different promoter polymorphisms within genes modulating inflammation have been demonstrated to elevate the AD risk. Tumor necrosis factor-alpha is a pro-inflammatory cytokine that has an essential role in the inflammatory process that is observed in Alzheimer's disease (AD) patient’s brain (17). Single nucleotide polymorphism in promoter region of TNF-a is among the important risk factor candidates. Studies in some population have shown the association of -308 G/A promoter polymorphism in TNF-a gene as a risk factor in Alzheimer’s disease. Thus it was decided to decipher the existence of any association between this polymorphism and Alzheimer’s disease in Iranian population.
In this study, 140 AD patients and 158 healthy controls were recruited. Statistical analyses showed no significant differences between AD patient’s alleles and genotypes in comparison to control subjects. The APOE genotypes of the subjects had been deter-mined in a previous study. So the patients and controls were stratified based on their APOE-e4 status; however no statistically significant differences in the -308G/A genotypes were observed between APOE-e4 positive and negative subjects.
The result of our study in Iranian popula-tion is in agreement with some studies in other part of the world. In a study in the USA there was no notable difference in allele or genotype frequencies for the TNF-?-308G/A between control and AD patient groups. There was not any difference the age of onset for dementia symptoms among carriers and non-carriers of A allele, either (13). As we did not have the onset age of the dementia symptoms in our study, we did not use this data in our statistical analysis. In a study in Italy (2006), no significant differences were observed between patients and healthy controls, where as carriers of A allele showed a significantly lower mean age at onset than non-carriers of this allele (12). In another study on Italian population (2008), also no positive associ-ation was found between this polymorphism and Alzheimer’s disease (17). Two studies in China have shown that this polymorphism is associated with the risk of LOAD and the frequency of A allele was significantly in-creased in AD patients compared to control subjects, (11, 18). These results are in contradict-tion with our findings. The -308 G/A poly-morphism was studied among Caucasian population and the frequency of A allele varied from 13% to 29% (6, 19); our result suggests that the frequency of A allele in Iranian population (9%) is lower than Cauca-sian population and is more comparable to Asian population based on Wang and col-leagues report (11). In conclusion, our result indicates a lack of association between -308 G/A polymorphism and risk of developing Alzheimer’s disease in Iranian population. However, a bigger sample size maybe needed for stronger confirmation of our findings.
Acknowledgement :
We express our sincere thanks and grati-tude to all Alzheimer's and control persons or their families for their kind participation in this study. We also thank Iran Alzheimer Association for their sincere collaborations.
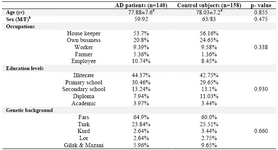
Table 1. Comparison of mean age, sex, occupations, education levels and genetic backgrounds between AD cases and control subjects using t-test and χ2 test analysis
a Mean±S.D.
b Male/ Female
|
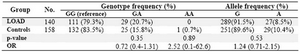
Table 2. The distribution of TNF-α-308G/A genotype and allele frequencies
|
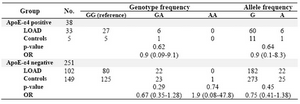
Table 3. Odd ratios for Alzheimer’s disease risk according to interaction of TNF-α genotypes with APOE-ε4 genotype
|
|