Hypoglycemic Effect of Aquatic Extract of Stevia in Pancreas of Diabetic Rats: PPARγ -dependent Regulation or Antioxidant Potential
-
Assaei, Raheleh
-
Endocrinology and Metabolism Research Center, Nemazee Hospital, Shiraz University of Medical Sciences, Shiraz, Iran
-
 Mokarram, Pooneh
Gasteroenterohepatology Research Center, Nemazee Hospital, School of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran. Department of Biochemistry, Faculty of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran, Tel: +98 71 32303029, E-mail: mokaram2@gmail.com, suny.respina@gmail.com
Mokarram, Pooneh
Gasteroenterohepatology Research Center, Nemazee Hospital, School of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran. Department of Biochemistry, Faculty of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran, Tel: +98 71 32303029, E-mail: mokaram2@gmail.com, suny.respina@gmail.com
-
Gasteroenterohepatology Research Center, Nemazee Hospital, School of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran
-
Department of Biochemistry, Faculty of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran
-
 Dastghaib, Sanaz
Endocrinology and Metabolism Research Center, Nemazee Hospital, Shiraz University of Medical Sciences, Shiraz, Iran, Tel: +98 71 32303029, E-mail: mokaram2@gmail.com, suny.respina@gmail.com
Dastghaib, Sanaz
Endocrinology and Metabolism Research Center, Nemazee Hospital, Shiraz University of Medical Sciences, Shiraz, Iran, Tel: +98 71 32303029, E-mail: mokaram2@gmail.com, suny.respina@gmail.com
-
Endocrinology and Metabolism Research Center, Nemazee Hospital, Shiraz University of Medical Sciences, Shiraz, Iran
-
Department of Biochemistry, Faculty of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran
-
Darbandi, Sara
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Darbandi, Mahsa
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Zal, Fatemeh
-
Reproductive Biology Group, Graduate School of Biomedical Sciences, Shiraz University of Medical Sciences, Shiraz, Iran
-
Akmali, Masoumeh
-
Department of Biochemistry, Faculty of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran
Abstract: Background: Traditional medicines with anti-diabetic effects are considered suitable supplements to treat diabetes. Among medicinal herbs, Stevia Rebaudiana Bertoni is famous for its sweet taste and beneficial effect in regulation of glucose. However, little is known about the exact mechanism of stevia in pancreatic tissue. Therefore, this study investigated the possible effects of stevia on pancreas in managing hyperglycemia seen in streptozotocin-induced Sprague-Dawley rats.
Methods: Sprague-Dawley rats were divided into four groups including normoglycemic, diabetic and two more diabetic groups in which, one was treated with aquatic extract of stevia (400 mg/kg) and the other with pioglitazone (10 mg/kg) for the period of 28 days. After completion of the experimental duration, rats were dissected; blood samples and pancreas were further used for detecting biochemical and histopathological changes. FBS, TG, cholestrol, HDL, LDL, ALT and AST levels were measured in sera. Moreover, MDA (malondialdehyde) level, catalase activity, levels of insulin and PPARγ mRNA expression were also measured in pancreatic tissue.
Results: Aquatic extract of stevia significantly reduced the FBS, triglycerides, MDA, ALT, AST levels and normalized catalase activity in treated rats compared with diabetic rats (p<0.05). In addition to this, stevia surprisingly, increased PPARγ and insulin mRNA levels in treated rats (p<0.05). Furthermore, stevia compensated for the histopathological damage in diabetic rats.
Conclusion: It is concluded that stevia acts on pancreatic tissue to elevate the insulin level and exerts beneficial anti-hyperglycemic effects through the PPARγ-dependent mechanism and stevia’s antioxidant properties.
Introduction :
Diabetes as a common chronic disorder of carbohydrates is the seventh leading cause of death in the world which needs constant care and attention 1. Complications in diabetes can be decreased by various approaches including diet, exercise and pharmacologic therapy. In modern medicine, the beneficial effects of drugs on glycemic levels are well documented but their preventive activity against progressive nature of diabetes and its micro and macro-vascular complications are not always effective. Moreover, synthetic antioxidants are often suspected to be carcinogenic. Therefore, herbal plants are preferred because of cost, having fewer side-effects and anti-oxidant activities. Stevia rebaudiana bertoni as an herbal plant is famous due to its sweet taste and beneficial effects in blood glucose regulation. In Japan and Brazil, extract of stevia is used as food additives and non-caloric sweeteners. Stevia leaves contain eight glycosides including dulcosides A, rebaudiosides A-E, stevioioside and stevioside. Stevioside is the sweetest glycoside found in stevia with antioxidant, antimicrobial, and anticancer properties 2-5.
Currently, researchers have shown that the treatment of hyperglycemia would be facilitated by the use of anti-diabetic compounds such as stevia, Psacalium peltatum, Curcubita ficifolia, Lepechinia caulescens due to their antioxidant properties. Moreover, scientists showed that anti-diabetic compounds with anti-oxidant properties would be more beneficial in treatment of diabetes 6-8. There are several approaches to target decreasing glucose including GLUT 2, glucokinase, lactate dehydrogenase levels and pyruvate carboxylase activity, mitochondrial glutamate efflux, ATP-sensitive K+ (K+ATP) channels, L-type voltage-dependent calcium channels and PPARγ 9,10.
Peroxisome proliferator-activated receptor-γ (PPARγ) is a nuclear hormone receptor that maintains homeostasis of glucose by activating glucokinase and glucose transporter 2 (GLUT2) in the liver and pancreas. Studies have indicated strong correlations between PPARγ and anti-diabetic action of thiazolidinediones (TZDs) such as pioglitazone which is a common anti-hyperglycemic drug in patients with diabetes. In this regard, researcher showed that Punica granatum flower (PGF) also exerts its anti-diabetic effects via PPARγ 11. In addition, PPARγ has insulin-sensitizing effects in peripheral tissues as well as the ability to sense blood glucose in pancreatic β-cells 10,12,13.
Moreover, it has been reported that Stevia rebaudiana shows the ability to maintain blood glucose level with glucose tolerance enhancement in diabetic rats 7. Stevia could also cause hypoglycemia in patients with diabetes through decreasing glycogenolysis and gluconeogenesis and absorbing glucose in the duodenum. In addition, antihyperglycemic and antioxidative potential of stevia and its glycoside were detected in several tissues such as kidney, liver and pancreas 14-16.
To our knowledge, the exact hypoglycemic mechanism of stevia and the role of PPARγ are not fully understood in pancreas. The current research was an attempt to evaluate whether stevia may improve glucose sensitivity of pancreatic cells as well as insulin secretion through the PPARγ- dependent mechanism.
Materials and Methods :
Chemicals: Streptozotocin and Pioglitazone powder was provided by Sigma (Germany) and pharmaceutical companies (Osveh, Iran), respectively. Stevia leaves were purchased from Golsaran Company, Rasht, Iran. TBA (thiobarbituric acid), TEP (1, 1', 3, 3'-tertaethoxy propane) were purchased from Sigma (Germany). Primers were provided by Methabion (Germany). RNA later solution and cDNA synthesis kit were purchased from Qiagen (USA). Syber green mix was provided by ABI Company (USA).
Animals: Adult male Sprague-Dawley rats (4 months, 250±20 gr) were procured from Shiraz University of Medical Sciences. All rats were acclimatized to new environmental conditions for one week, prior to initiation of experiment. They were housed under standard laboratory conditions of 12 hr light and 12 hr dark cycle, fed with standard diet; chow diet (Pars Dam Co, Tehran, Iran) and water ad libitum. All experimental rats were divided into 4 groups each containing 10 rats. Maintenance and treatment of all the animals were done in accordance with the principles of Institutional Animal Ethics.
Experimental design: Diabetes is induced in mice by using a single intraperitoneal injection of Streptozotocin with a dose of 40 mg/kg body weight in 10 mmol/l sodium citrate buffer with pH=4.5 in overnight fasted rats 17.
Following the diabetes induction (after 5 days), blood was drawn from the tail vein of experimental rats to determine their fasting blood glucose levels. Rats showing fasting blood sugar greater than 300 mg/dl or more, developed diabetic symptoms such as weight loss, hyperplasia, polydipsia and polyuria. These were considered diabetic and selected for the treatments using either pioglitazone (10 mg/kg) or aquatic extract of stevia (400 mg/kg) 18.
Our cohort comprised the following groups (i) Group A: water, 1ml (Control); (ii) Group B: water, 1 ml (diabetic control); (iii) Group C: 10 mg/kg pioglitazone,1 ml; (iv) Group D: 400 mg/kg aquatic extract of stevia, 1 ml. All groups received the treatment by gavage in a single dose every morning for 28 days.
Preparation of aquatic extract of stevia: Stevia leaves were procured from Rasht University of Agriculture and Technology, Gilan. Dried leaves were subjected to size reduction like coarse powder. Subsequently, the powder was soxhlet extracted and evaporated to dryness under reduced pressure in vacuum rotatory evaporator. The extract was air dried till solid to semisolid mass was obtained. The suspension of 100 g of powder extract was prepared in 1200 ml of distilled water and kept in dark condition 19,20.
The evaluation of Stevioside in Stevia leaves: Stevioside concentration was measured by Macaulay et al’s method 21. Filtered solution of stevia extract was injected to HPLC Agilent system. Stevioside (2000 mg/l with 95% purity) was used as the standard solution. According to the Area Under the Standard Peak (AUSP), stevioside concentration in the stevia plant leaves was determined (Figure 1).
Serum glucose, liver enzyme and lipid profile analysis: For estimating serum glucose, lipid profile, and amino tranferases (ALT, AST), serum was isolated from the blood collected from overnight fasted rats after 28 days of treatment. Serum glucose, triglyceride, cholesterol, HDL, LDL, ALT, AST were measured after 28 days of treatment using diagnostic colorimetric kits (BioSystem, Spain) by prestige instrument (Hitachi Japan).
Preparation of RNA and homogenate from pancreas: Pancreas was removed and washed fast with ice cold normal saline solution (0.9%). Pancreas was divided into two parts for estimating several biochemical factors as well as molecular analysis. The former was snap frozen and rapidly cut into small pieces and homogenized in 5 ml cold-normal saline with the help of homogenizer (Potter Elvehjem, Bodine Electric and Chicago, IL, USA). The homogenate was filtered and then centrifuged at 10,000 g for 1 hr at 4oC. The supernatant obtained was used for estimation of total protein, malondialdehyde (MDA) and catalysis of enzyme activity.
The latter was immersed into RNA later solution (Qiagen, USA) and total RNA was extracted based on our recent procedure 22 to evaluate the levels of PPARγand insulin mRNAs expression.
Determination of lipid peroxidation marker and catalase activity: Malondialdehyde (MDA) as the representative of lipid peroxidation level was measured using Hagar et al’s method 23. In brief, MDA was provided as an acetal derivative because the aldehyde itself is not stable. The acetal form (TEP or 1, 1', 3, 3'-tertaethoxy propane) was hydrolysed during the acid incubation step at 95oC and generated MDA. The TEP standard was provided as a 10 mM stock solution in Tris-HCL, and diluted 1/500 (v/v) in water. The MDA concentrations were determined according to a standard curve at 532 nm.
Catalase was also assayed spectrophotometrically by the Aebi's procedure based on the content of H2O2 which degraded 24. Briefly, 66.7 mmol/l phosphate buffer (pH=7.0), 20λ of pancreas supernatant and 355λ distilled water were added to 75 ul of H2O2 (120 mmol/L) and the consumption of H2O2 was followed spectrophotometrically at 240 nm for 3 min at 25oC. Catalase activity was expressed as μmol H2O2 consumed/min per mg pancreas supernatant protein using a molar extinction coefficient of 43.6 l/mol per cm for H2O2.
Evaluation of insulin and PPARγ mRNA expression in pancreases by semi-quantitative RT-PCR: Pancreatic tissues were immersed into RNA later solution (Qiagen, USA) for 24 hr and kept in -80°C, then, total RNA was extracted from 50-100 mg of tissues based on our recent procedure 22 and followed with Tripure Isolation Reagent (Qiagen, USA).
Finally, cDNA was synthesized from 1 μg of total RNA by QuantiTect Reverse Transcription Kit (Qiagen, USA). The reverse transcription was performed in a 20 μl reaction volume system. PCR reaction system of 25 μl contained 2 μl of cDNA, 1 μl 10 pmol forward and reverse primers (sequences of primers in table 1), 2.5 μl of 10 X PCR buffer, 0.5 μl of dNTP mixture (each 2.5 mM) and 1.5 μl Mgcl2 and 0.3 μl Taq DNA polymerase (5 U/μl). PCR was performed at 95oC for 5 min followed by 35 cycles of 95oC for 30 s, 57oC for 30 s and 72oC for 45 s and then 72ºC for 10 min. The PCR products were separated on a 2% (w/v) agarose gel and visualized by gel red staining.
226 and 209 bp amplicons were expected upon performing PCR for PPARγ and insulin, respectively. PCR primers for the analysis of rat β actin mRNA level as an internal control and PPAR-γ and insulin are indicated in table 1. Primers for internal control also result in a 138 bp amplicon.
Quantitative real time PCR: Real time PCR was carried out using the ABI real time PCR 7500 system. The PCR reac¬tion mixture contained 2 μl of cDNA (tenfold diluted), 0.5 μL of 5 mmol/l solutions of each of the forward and reverse primers and 10 μl of SYBR green DNA PCR Master Mix in a total volume of 20 μl. All incubations included an initial denaturation step at 95°C for 10 min and 40 cycles (15 s at 95°C and 30 s at 60°C) subsequently. A melting curve analysis was achieved by performing 70 cycles of 10 s with a temperature increment of 0.5°C/cycle starting from 60°C. Efficiency of amplification was measured by the slope of a standard curve. Data were analyzed by using the 7500 Software v 2.0.1. The relative expression level of insulin and PPARγ genes was calculated by the 2-ΔΔCT formula 25,26. β Actin was also considered as an internal control.
Histological study: Pancreas of all experimental groups was further studied for their histopathological changes. Parts of pancreatic tissue were fixed in 4% formalin. The organs were then processed in graded series of alcohol and embedded in paraffin wax at 55°C in an incubator to form a block. Serial sections of 5 μm were cut using microtome. Ribbons formed were then mounted on glass slide, passed again through series of alcohol and stained with hematoxylin-eosin and photomicrographed at 400 times magnification 27.
Immunohistochemical study of pancreatic islets tissue for evaluation of insulin granules: Four-micrometer sections were prepared from paraffin-embedded pancreas blocks for IHC analysis 28. Briefly, each section was deparaffinized in xylene and rehydrated in alcohol. Antigen retrieval was performed in 10 mM citrate buffer (pH=6.0) at 122°C for 3 min (using an electric pressure cooker). Specimens were subsequently blocked and incubated further with a mouse monoclonal antibody raised against rat insulin (ready to use; Dako, Denmark Cat.No.N1542).
Antigen-antibody reactions were revealed using a commercial kit (Dako, Denmark Cat.No.QD620-YIK) at room temperature; diaminobenzidine (DAB) was used to detect the expression signals. Finally, the sections were counterstained with hematoxylin. Furthermore, positive and negative controls were appropriately used.
Statistical analysis: Results are expressed as mean±standard error of mean (SEM). Statistical analysis was performed using one-way Analysis of Variance (ANOVA) followed by Tukey’s post-hoc multiple comparison tests using SPSS (Version 16; SPSS, Chicago, IL, USA). The values of p<0.05 were considered statistically significant.
Results :
The present study was designed to elucidate the possible mechanism of aquatic extract of stevia in managing the sugar level via PPARγ and insulin in pancreas tissue.
As shown in table 2, there is no significant difference between all variants in groups B, C, D at the baseline, whereas some of our variants were significantly changed after 28 days in the individual groups compared with baseline as we had expected.
Body weight: Body weight significantly decreased (p<0.001) in all diabetic groups when compared with control one. Pioglitazone (group C) significantly increased weight compared with diabetic group (p<0.05). However, stevia significantly decreased weight compared with 10 mg/kg pioglitazone group (p<0.05) (Table 3).
Serum glucose and lipid profile: FBS significantly increased in all diabetic groups when compared with control rats (p<0.001). Surprisingly, groups treated with either pioglitazone or stevia showed significant decrease in blood glucose compared to diabetic group. However, 400 mg/kg of stevia was more effective than 10 mg/kg of pioglitazone in lowering glucose (Table 3). No differences were seen in amount of cholesterol in diabetic group compared with control one. Overall, it seems that stevia could also improve the amount of serum TG compared with diabetic and pioglitazone groups (Table 3).
Serum transaminase activity: Groups treated with either stevia or pioglitazone (group C or D) showed significant decrease in the liver enzymes activity compared with diabetic control rats (p<0.05). There is no difference in serum ALT and AST between group C and D (Table 3).
Pancreas catalase activity: As shown in table 3, activity of catalase in homogenate of pancreas significantly decreased in diabetic rats compared with control (p<0.001) whereas activity of the enzyme increased in the groups treated with stevia or pioglitazone compared with control and diabetic rats, respectively (p<0.001).
Pancreas lipid peroxidation: The results showed that groups treated with either stevia or pioglitazone could normalize the amount of MDA compared to diabetic group (Table 3).
Analysis of PPARγ and insulin mRNA expression: The presence of appropriate bands for PPARγ (224 bp), insulin (209 bp) and β actin (138 bp) amplicons is shown in figures 2 and 3.
Density of PPARγ and insulin bands was normalized with syngen software (Figures 4 and 5). Since PPARγ expression changes in groups treated with stevia and pioglitazone compared to control were not statistically significant, further investigation was done by real time PCR quantitation. The analysis of data showed that pioglitazone and stevia significantly increased PPARγ levels and insulin mRNA genes in the treated group compared with diabetic ones (p<0.05) (Figure 6).
Pancreas histopathological effect: The Langerhans islet beta cells were degenerated, vacuolated in diabetic rats and the number of islets significantly decreased. Regeneration of islets was determined in groups treated with stevia, pioglitazone (Figure 7).
Determination of insulin granules of pancreas tissue by immunohistochemistry: The results of IHC for insulin granules were semi-quantitated using the evaluation of positive cells. According to figure 8, pioglitazone and aquatic extract of stevia have relatively better effect on synthesis and aggregation of insulin in the pancreas. However, none of them could reach the granules of islet cell as much as control group.
Discussion :
Nowadays, hypoglycemic medications are attractive for treatment of diabetes. Herbal plants such as Teucrium polium, Smallantus sonchifolius (yacon), Psacalium peltatum, etc, have been proposed to have hypoglycemic effects 8,29-31. Our study also showed that aquatic extract of stevia has beneficial anti-diabetic effects in decreasing the glucose triglycerides, MDA, ALT, AST, whereas it could not improve weight loss in diabetic rats.
The role of stevia in decreasing the glucose level was evaluated by focusing on the PPARγ and insulin levels in pancreas. Levels of insulin and PPARγ mRNA expression were also significantly increased in diabetic groups that were treated either with stevia or pioglitazone. Stevia also compensated the histopathological damage and elevated level of insulin in granules in diabetic rats.
During diabetes or insulin resistance, failure of insulin-stimulated glucose uptake by fat and muscle causes high glucose concentration in blood. Consequently, glucose uptake by insulin-independent tissues, oxidant production and impaired antioxidant defenses occur. Increased oxidative glucose metabolism itself increases mitochondrial production of the superoxide anion (O2-), which will be converted to the hydroxyl radical (OH-), and hydrogen peroxide (H2O2).
Among biological targets of oxidative stress, lipids are the most involved class of biomolecules. Furthermore, MDA is the principal and most studied product of polyunsaturated fatty acid peroxidation which plays important role in etiopathology of several chronic diseases including diabetes 32.
In this regard, antioxidant compensates the action of free radicals by several mechanism including: 1) enzymes that degrade free radicals such as super oxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px)….. 2) proteins such as transferrin that can bind metals which stimulate the production of free radicals, and 3) antioxidants such as vitamin C and E that act as free radical scavengers 33.
Therefore, imbalances of oxidative stress/antioxidant defense system are important factors in patients with diabetes 6,7 and it seems that the aquatic extract of stevia could decrease the amount of MDA and improve catalase activity as indicator of oxidative stress and antioxidant defense system, respectively. A clear correlation was also observed between the catalase activity and lipid peroxide concentrations in the diabetic rats. These observations suggest that there are similar abnormalities in lipid metabolism and pancreatic antioxidant enzymes in diabetic rats 33. As shown in table 3, elevated levels of lipid peroxidation products (MDA) were found in diabetic rats. This might be due to overproduction of peroxides and inhibition of activities of peroxidases which is a common event in diabetic rats. Reduction of peroxides formation led to lower formation of MDA in group D (Table 3) and it corresponded to a restored activity of catalase. Consistent with the results of the current study, the anti-oxidant effects of stevia has been evaluated recently. The result showed that the root extract of stevia has also significant anti-oxidative potential 15. In addition, similar to other reports 34-36, ALT and AST levels decreased in diabetic rats after being treated with Iranian breed of stevia. Therefore, anti-oxidant effects of stevia on pancreas and its hepato-protective properties can be used in the management of oxidative stress caused by the disease.
On the other hand, oxidative stress along with hyperglycemia plays a major role in the pathogenesis of diabetes. Stevia has antihyperglycemic and anti-oxidative potential due to high phenols and flavanoid contents 15. To our knowledge, the presence of some biomolecules present in the stevia may stimulate the beta cells to release insulin, leading to improvement in the carbohydrate metabolizing enzymes and thus establishing normal blood glucose level.
Although stevia decreased glucose level in this study, it could not normalize body weight after 28 days. According to previous study, it might also act as weight loss agent 37. Therefore, the effect of stevia on weight should be considered as one limitation during treatment with stevia.
Another factor that influences glucose hemostasis is PPARγ, a member of the ligand-activated nuclear receptor superfamily. It involves in several biological processes, such as inflammation, glucose metabolism, angiogenesis and adipogenesis. Consequently, stimulation of peripheral glucose utilization and enhancing glycolytic could be an alternative mechanism in decreasing glucose 10. Furthermore, severe hyperglycemia is seen in patients that have a dominant-negative mutation in the PPARγ gene. Level of PPARγ mRNA also declined in rats treated with a high-fat diet. Adipose-specific PPARγ null mice also have higher levels of circulating lipids, increased hepatic gluconeogenesis, and insulin resistance. In addition, the presence of PPARγ response elements in the promoter of GLUT-2 and β-glucokinase genes has an important role in regulating insulin release from the pancreas 38,39.
In this regard, our quantitative real time PCR data showed that level of mRNA PPARγ was significantly reduced in diabetic group compared with normoglycemic rats. Our data also showed the PPARγ mRNA level increased in groups treated with stevia or pioglitazone.
Some studies showed that there is a strong correlation between the TZD- PPARγ interaction and anti-diabetic action of TZDs 40; it seems that the relative potency of TZDs for binding to PPARγ and activation of PPARγ in vitro associates well with their anti-diabetic potency in vivo.
To our knowledge, the increased level of PPARγ in our treated group could induce the insulin secretion and control the level of blood glucose in diabetic rats. In this respect, Ferreira et al demonstrated that the activity of PPARγ is increased when U937 cells are co-transfected with expression vector encoding the PPARγ receptor in presence of pioglitazone as an anti-oxidant agent 41. Furthermore, Atten et al found that anti-oxidants up-regulate PPARγ and decrease fibrosis in chronic pancreatitis 42. On the other hand, high PPARγ expression ratio in our treated rats marked that stevia and pioglitazone can act as similar ligands on PPARγ to increase the release of insulin from the pancreas. However, further study such as affinity binding assay on stevia and pioglitazone is needed to answer whether stevia mimics the pioglitazone role to interact with the PPARγ.
On the other hand, stevia and pioglitazone could significantly increase the level of insulin protein in pancreatic tissue. Our findings demonstrated that stevia similar to other anti-diabetic plants such as bitter melon or Karela (Momordica charantia), Chard (Beta vulgaris L.) and Teucrium polium L. (Labiatae) increased significantly the level of insulin granules in IHC experiment 28,43,44. Therefore, it seems that aquatic extract of stevia could act as insulinotropic agent and our data is consistent to other results which demonstrated stevioside and steviol as derivatives of stevia which enhance insulin secretion by directly acting on β-cells during in vitro analysis 45-47.
According to our results, stevia can improve the glucose concentration via several mechanisms. Stevia increases the level of insulin mRNA more than the level of PPARγ mRNA in diabetic rats PPARγ. It also seems that anti-oxidant effect of stevia on the pancreas is an additional mechanism to increase the release of insulin more than PPAR levels. Other explanation is related to the rise in PPARγ which is enough to induce insulin secretion.
Although stevia could act as an antihyperglycemic agent due to the ability to restore the function of pancreatic tissues by causing an increase in insulin output via PPARγ, the antihyperlipidemic effect of stevia is still attractive for our research team. Because stevia could not change the level of cholesterol, LDL, and HDL compared to TG in sera in our diabetic groups, which is inconsistent with other studies 48. This discrepancy could be attributed to our limitation in establishing an appropriate diabetic model to show alteration in lipid profile. In addition, the low dose of streptozotocin and short duration time could be one related limitation which should be considered in future studies. In addition, it is very important to design in vivo in combination with in vitro studies for a better insight into the anti-diabetic actions of stevia that could be subjected to the clinical trial.
In general, it can be concluded that beside antioxidant effect of stevia in pancreas, stevia acts on β-cell and increases the level of PPARγ and insulin mRNAs. However, further studies including gene array analysis, protein assay of PPARγ with western blot, and stereology are needed to elucidate the possible mechanism of stevia in main tissues including liver, pancreas, and peripheral tissues for management of diabetes in the clinic.
Conclusion :
It is concluded that stevia acts on pancreatic tissue to elevate the insulin level and exerts beneficial anti-hyperglycemic effects through the PPARγ-dependent mechanism and stevia’s antioxidant properties.
Acknowledgement :
The present article was extracted from the thesis written by Sanaz Dastghaib and financially supported by Shiraz University of Medical Sciences Grant No, 91-6137.
Conflict of Interest :
The authors declare that they have no competing interests. All authors read and approved the final manuscript.

Figure 1. The area under the standard peak (AUSP). Stevioside standard (A) and aquatic extract of stevia (B).
|
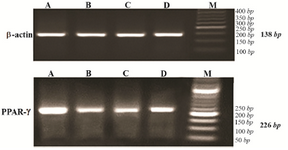
Figure 2. Agarose gel electrophoresis of RT-PCR products of PPARγ expression (226 bp) and β actin as internal control (138 bp) for experimental groups, stained with gel red. A) control group; B) diabetic group; C) diabetic rats received 10 mg/kg pioglitazone; D) diabetic rat received 400 mg/kg aquatic extract of stevia.
|
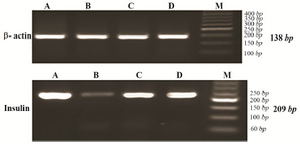
Figure 3. Agarose gel electrophoresis of RT-PCR products of insulin expression (209 bp) and β actin as internal control (138 bp) for experimental groups, stained with gel red. A) control group; B) diabetic group; C) diabetic rats received 10 mg/kg pioglitazone; D) diabetic rat received 400 mg/kg aquatic extract of stevia.
|
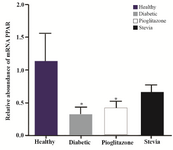
Figure 4. PPARγ expression status in four experimental groups. The PPARγ expression was determined using syn-gen software. Data are presented as mean±standard error of the mean (SEM). Sample size (n=8). *p<0.05 for significant changes compared with control.
|
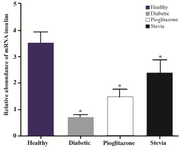
Figure 5. Insulin expression status in four experimental groups. Insulin expression was determined by syn-gen software. Data are presented as mean±standard error of the mean (SEM). Sample size (n=8). *p<0.05 for significant change compared to control.
|
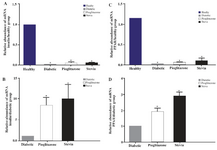
Figure 6. The PPARγ and insulin mRNA expression were measured by real time PCR and analyzed with the 2 ΔΔCT method. Comparisons between different treated groups were performed using One Way ANOVA. Part A, C compared to control and B, D compared to diabetic rats for PPARγ and insulin mRNA expression. Values are mean±SEM (n=5) *p<0.05.
|
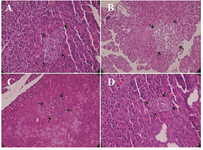
Figure 7. Representative histopathological profiles on β-cells (arrows show one islet) in the A) control B) diabetic, C) diabetic rats treated with pioglitazone and D) diabetic rats treated with stevia (H&E ×400).
|
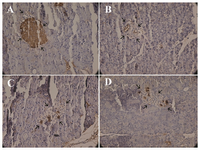
Figure 8. Insulin-immunoreactive granules (show with arrow) in β-cells. A) control B) diabetic, C) diabetic rats treated with pioglitazone and D) diabetic rats treated with stevia (×400).
|
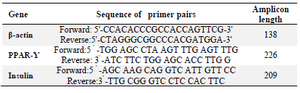
Table 1. Sequences of primers
|
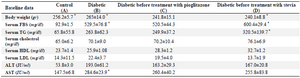
Table 2. Baseline data before treatment
Results are presented as mean±SEM (n=12), differences between values in baseline and the end of treatment in individual groups. *p<0.05
|
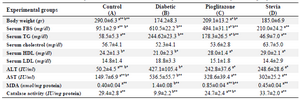
Table 3. The effect of aquatic extract of stevia on biochemical parameters in different experimental groups
Results are presented as mean±SEM, n=12. a) Compared with diabetic groups. b) Compared with stevia groups. *p<0.05, **p<0.01
|
|