Antinociceptive and Antipyretic Activities of Amaranthus Viridis Linn in Different Experimental Models
-
 Srinivas Ashok Kumar, Bagepalli
B.S.,M. Pharm (Ph.D.), Department of Pharmacognosy,Sri K.V.College of Pharmacy, Chickballapur, Karnataka, India, E-mail: ashok4vani@gmail.com
Srinivas Ashok Kumar, Bagepalli
B.S.,M. Pharm (Ph.D.), Department of Pharmacognosy,Sri K.V.College of Pharmacy, Chickballapur, Karnataka, India, E-mail: ashok4vani@gmail.com
-
Department of Pharmacognosy, Sri K.V.College of Pharmacy, Chickballapur , Karnataka, India
-
Lakshman, Kuruba
-
Department of Pharmacognosy, PES College of Pharmacy, Bangalore , Karnataka, India
-
Konta Narsimha Jayaveera, Korala
-
Department of Chemistry, Jawaharlal Nehru Technological University of College of Engineering, Anantapur , Andhra Pradesh, India
-
Manoj, Bachappa
-
Department of Pharmacognosy, Sri K.V.College of Pharmacy, Chickballapur , Karnataka, India
Abstract: Methanolic extract of whole plant of Amaranthus viridis L (MEAV), was screened for antinociceptive activity using acetic acid induced writhing test, hot plate test and tail immersion test in mice. In a similar way a screening exercise was carried out to determine the antipyretic potential of the extract using yeast induced pyrexia method in rats. Administration of the extracts was applied to both laboratory animals at the doses of 200 and 400 mg/kg body weight, respectively. The results of the statistical analysis showed that MEAV had significant (p<0.01) dose dependent antinociceptive and antipyretic properties at 200 and 400 mg/kg. Hence present investigation reveals the antinociceptive and antipyretic activities of methanolic extract of Amaranthus viridis.
Introduction :
Amaranthus viridis L (A. viridis Amaran-thaceae), commonly called ‘Chilaka Thota-Kura’ in Telugu, has been used in Indian and Nepalese traditional system to reduce labour pain and act an antipyretic (1,2). The Negritos of the Philippines apply the bruised leaves directly to eczema, psoriasis and rashes etc (3). Other traditional uses range from an anti-inflammatory agent of the urinary tract, venereal diseases vermifuge, diuretic, anti-rheumatic, antiulcer, analgesic, antiemetic, laxative, improvement of appetite, antilep-rotic, treatment of respiratory and eye prob-lems, to treatment of asthma (1,4-11).
Furthermore, the plant possesses antiprolif-erative and antifungal lactin properties as well
as ribosome inactivating protein, ß-carotene (12-14) and antiviral activities (15). In addition the whole plant possesses analgesic and anti-pyretic properties and is used for the treat-ment of pain and fever respectively in trad-itional systems of medicine (16). However, there is not enough scientific reports to sup-port these supposed analgesic and antipyretic activities. This has prompted us to conduct the studies to ascertain the authenticity of these important claims of traditional potency.
Materials and Methods :
Collection and Extraction of Plant Material
Fresh plant material of A. viridis was col-lected from surroundings of Chickballapur, Karnataka in the month of May 2009. The plant material was identified and authentic-ated by Dr. Rajan, Department of Botany, Government Arts College, Ootcamund, Tamilnadu.
A voucher specimen (SKVCP 13) was de-posited in college herbarium. Plant material was washed with water to remove dirt and shade dried for one week. The dried material was powdered by using grinder and passed through 10-mesh sieve. The coarsely pow-dered material was extracted with methanol by using soxhlet apparatus. The extract was later evaporated to dryness under reduced pressure and the residue was preserved for future use.
Preliminary phytochemical screening
The methanol extract of A. viridis was screened for the presence of various phyto-constituents like steroids, alkaloids, glycol-sides, flavonoids, carbohydrates, proteins and phenolic compounds (17).
Animal models
Male Swiss albino mice weighing 20-25 g were acclimatized to the experimental room at temperature 23±2°C, controlled humidity con-ditions (50-55%) and 12 hr light and 12 hr dark cycle. A maximum of two animals were kept in a polypropylene cage and fed with standard food pellets (Kamadenu Enterprises, Bangalore) and water ad libitum.
Acute toxicity studies
Methanol extracts of A. viridis was studied for acute oral toxicity as per revised OECD (Organization for Economic Cooperation and Development) guidelines No. 423. The extract was devoid of any toxicity in rats when given in doses up to 2000 mg/kg by oral route. Hence, in our study 200 and 400 mg/kg doses of extract were dissolved in 0.1% Carboxy Methyl Cellulose (CMC) and used for the study (18).
Antinociceptive activity
Acetic acid-induced writhing test: This test was done using the method described by Collier et al (19). Muscle contractions were induced in mice by intra peritoneal injection of 0.6 % solution of acetic acid (10 ml/kg). Thirty minutes before this administration the animals were treated with diclofenac sodium (50 mg/kg), MEAV orally at doses of (200 and 400 mg/kg) and 0.1 % CMC (5 ml/kg). Immediately after administration of acetic acid, the animals were placed in glass cages, and the number of ‘stretching’ per animal was recorded during the following 15 min.
Writhing movement was accepted as con-traction of the abdominal muscles accom-panied by stretching of hind limbs. There was significant reduction in the number of writhes by drug treatments as compared to vehicle treated animals. This was considered a posi-tive analgesic response and the percentage inhibition of writhing was calculated (19).
Hot plate method: The hot plate test described by Eddy and Leimback (1953) was used. The mice were first treated with differ-ent doses of MEAV (200 and 400 mg/kg orally). One hour after this administration the animals were placed on a hot plate maintained at 55±1.0 °C. A cut-off period of 15 sec was considered as maximal latency to avoid injury to the paws. The time taken by the animals to lick the hind paw or jump out of the place was taken as the reaction time and was measured at 0,30,60 and 120 mins. Morphine (5 mg/kg) was used as a reference drug (20).
Tail immersion: Tail immersion was con-ducted as described by Aydin et al (21). This involved immersing extreme 3cm of the rat’s tail in a water bath containing water main-tained at a temperature of 55±0.5°C. Within a few minutes, the rats reacted by withdrawing the tail. The reaction time was measured at 0, 30,60,120,180,240 and 300 mins.
The test groups were given MEAV (200 and 400 mg/kg), morphine (5mg/kg) and 0.1% CMC in water were administered orally (21).
Screening for antipyretic activity
The antipyretic activity of MEAV was evaluated using Brewer’s yeast-induced pyrexia in rats as described by Loux et al (22). Fever was induced by administering 20 ml/kg of 20% aqueous suspension of Brewer’s yeast in normal sali
Result :
The present study was conducted to assess the antinociceptive and antipyretic properties of methanolic extract of A. viridis. The methods selected were chemical nociception in the test model of acetic acid-induced writh-ing and thermal nociception hot plate and tail immersion test. These methods were selected to evaluate both centrally and peripherally mediated effects of MEAV. The acetic acid induced abdominal constriction is believed to show the involvement of peripheral mechan-isms, whereas the hot plate and tail immersion tests are believed to do same by central mechanisms (23).
Discussion :
Preliminary phytochemical study indicated the presence of alkaloids, steroids, glycosides, flavonoids, phenolic compounds, terpenoids, proteins and carbohydrates which might be re-sponsible for the antinociceptive and anti-pyretic effects of the MEAV.
Flavonoids and phenolic compounds have been reported to have multiple biological effects such as anti-oxidant activity (26), antinociceptive activity in vivo (27,28), anti in-flammatory action (29,30), inhibition of platelet aggregation (31), inhibition of mast cell hista-mine release (32) and inhibitory action on ar-achidonic acid metabolism as demonstrated by in vitro and in vivo tests (33).
Acknowledgement :
The authors wish to thank Sri K.V. Naveen Kiran, Chairman, Sri K.V. College of Phar-macy, Chickballapur, Karnataka (India), for providing the research facilities to carry out this work successfully.
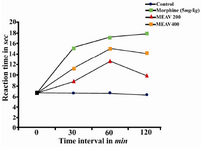
Figure 1. Effect of methanolic extract of Amaranthus viridis (MEAV) on hot plate test in mice
|
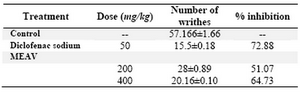
Table 1. Effect of methanolic extract of Amaranthus viridis (MEAV) on acetic acid induced writhing test in mice
Values are in mean �SEM; (n=6)
|
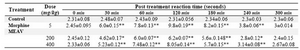
Table 2. Effect of methanolic extract of Amaranthus viridis (MEAV) on tail immersion test in mice
Values are in mean �SEM; (n=6) *p<0.05, ** p<0.01 vs control
|
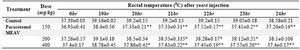
Table 3. Effect of methanolic extract of Amaranthus viridis (MEAV) on yeast induced pyrexia
Values are in mean �SEM; (n=6) *p<0.05, ** p<0.01 vs control
|
|