Comparison between Platinum-Azidothymidine and Azidothymidine Effects on Bcl-2 and Telomerase Gene Expression in Rats with Hepatocellular Carcinoma
-
Sabokrouh, Abdolreza
-
Department of Clinical Biochemistry, Hamadan University of Medical Sciences, Hamadan, Iran
-
 Vaisi-Raygani, Asad
Department of Clinical Biochemistry, School of Medicine, Kermanshah University of Medical Sciences, Kermanshah, Iran, Tel: +98 813 8380466; E-mail: asadvaisiraygani@kums.ac.ir
Vaisi-Raygani, Asad
Department of Clinical Biochemistry, School of Medicine, Kermanshah University of Medical Sciences, Kermanshah, Iran, Tel: +98 813 8380466; E-mail: asadvaisiraygani@kums.ac.ir
-
Molecular Diagnostic Research Center, Kermanshah University of Medical Sciences, Kermanshah, Iran
-
 Goodarzi, Mohammad Taghi
Research Center for Molecular Medicine, Hamadan University of Medical Sciences, Hamadan, Iran, Tel: +98 813 8380466; Email: mtgoodarzi@yahoo.com
Goodarzi, Mohammad Taghi
Research Center for Molecular Medicine, Hamadan University of Medical Sciences, Hamadan, Iran, Tel: +98 813 8380466; Email: mtgoodarzi@yahoo.com
-
Department of Clinical Biochemistry, Hamadan University of Medical Sciences, Hamadan, Iran
-
Research Center for Molecular Medicine, Hamadan University of Medical Sciences, Hamadan, Iran
-
Khatami, Shohreh
-
Department of Clinical Biochemistry, Pasteur Institute, Tehran, Iran
-
Shahabadi, Nahid
-
Department of Chemistry, Razi University of Kermanshah, Kermanshah, Kermanshah, Iran
-
Lakpour, Niknam
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Shakiba, Yadollah
-
Molecular Diagnostic Research Center, Kermanshah University of Medical Sciences, Kermanshah, Iran
Abstract: Background: High expression of telomerase and Bcl-2 are reported in hepatocellular carcinoma. Some anticancer drugs show their effects through reduction of these factors. In this study, it was aimed to investigate the effects of a new synthetic compound, platinum azidothymidine, on inhibition of telomerase and Bcl-2 expression in hepatocellular carcinoma compared to azidothymidine.
Methods: To study the effects of Pt-AZT on hepatocellular carcinoma and compare its effects with AZT in inhibition of telomerase and Bcl-2 gene expression, pathogen-free male Wistar rats (n=100) were used. They were randomly divided to 4 groups (n=25). Group A as the control group contained 25 healthy rats; in the rest of animals, preneoplastic lesions were induced in their livers (groups B, C, and D) using Solt-Farber resistant hepatocyte protocol. Cancer development was approved by a pathology laboratory. Group B was negative control (untreated), groups C and D were treated by intraperitoneal injection (IP) of Pt-AZT (0.9 mg/kg/day) and AZT (0.3 mg/kg/day), respectively for 14 days. At the end of the protocol, all rats were sacrificed and Bcl-2 and telomerase gene expression was determined using real -time PCR.
Results: No tumor in the livers was found in group A at any point of the study, but it was present in livers of all animals in B, C and D groups. Results showed that telomerase and Bcl-2 expression was significantly lower in group C compared with group B (0.473±0.231 vs. 5.137±5.08, p<0.001, for telomerase expression, and 0.41±0.276 vs. 7.25±11.6, p<0.001, for Bcl-2 expression) and also compared with group D (0.473±0.231 vs. 3.48±4.02, p<0.001, for telomerase expression, and 0.41±0.276 vs. 4.93±18, p<0.001, for Bcl-2 expression).
Conclusion: For the first time, it was demonstrated that Pt-AZT has more inhibitory effect on telomerase and Bcl-2 expression than AZT. It effectively inhibits the growth of liver tumor in rats by extending apoptosis.
Introduction :
Cancer is one of the major causes of mortality in the world and in 2008 accounted for approximately 7.6 million deaths (13% of all deaths). Cancer mortality rate is projected to increase to 11 million deaths in 2030 1,2. The prognosis of Hepatocellular Carcinoma (HCC) remains weak, because of its early metastasis and rapid progression. Medications used to treat this disease have not progressed so far and for this reason using stronger and more effective new drugs are required. In recent years, antiviral drugs and anticancer nucleosides to treat cancer were the center of focus e.g. Ribavirin and Azidothymidine (AZT) 3,4. The reviews in this field indicate an increase in the expression of telomerase and Bcl-2 in cancer cells 5,6.
Telomeres are tandem repeated guanines of rich sequences of 5′TTAGGG3′ at the end of chromosomes of all vertebrates which protect the ends of chromosomes from damage and degradation 7,8. Telomerase is a ribonucleoprotein enzyme and its expression extends telomeres and prevents DNA damage leading to maintenance of telomeres. Telomerase over expression was reported in a number of human cancer tumors 9,10. While in normal hepatocytes there are low telomerase activities, cancer cells including HCC cells contain high telomerase activity 11-13. For this reason, telomerase inhibition by some drugs is a novel approach for cancer therapy 14.
B cell lymphoma protein-2 (Bcl-2), a 26 kDa oncoprotein, is present in the outer mitochondrial membrane and poses antiapoptotic properties. By regulation the pores of mitochondria membrane it prevents release of caspase-3 activating factor that contributes to apoptotic pathway and therefore inhibits apoptosis. In normal cells, Bcl-2 expression is very low and in this condition Bcl-2 is known to promote cell survival, even when the cell proliferation rate is not elevated, it acts as the negative regulator. On the other hand, in cancer cells, the increase in both of its mRNA and protein level leads to resistance to apoptosis 15. However, Bcl-2 protein expression has been reported to increase in a variety of human cancer and its precise biological role in the development of malignant tumors is still controversial 16-21. Nevertheless, telomerase and Bcl-2 are potential targets for cancer therapy and their inhibition created a new approach in this field 14,22.
AZT inhibits synthesis of cancer genome by its active anabolite AZTTP (azidothymidine triphosphate) through chain termination mechanism which may inhibit telomerase activity competitively 23. Also, according to the pathological findings, treatment with AZT leads to a decrease in expression of genes such as telomerase and Bcl-2. AZT acts to arrest the cells with inducing senescence and apoptosis in tumor cells 24,25. High telomerase expression is present in 85-90.9% of tumor cells and telomerase activity is observed during the period that normal cells transit towards tumor cells 26. AZT interrupting reverse transcriptase of cells blocks the cell cycle, and inhibits replication of cells and cell growth 27,28; also, AZT inhibits several kind of enzymes in tumor cells and some of them contribute to cell cycle regulation such as Mad1. Consequently, reduction of these cell cycle factors inhibits cell growth in S phase and cells enter apoptosis phase which indicates AZT is an effective anticancer drug 29,30.
There is no published study showing in vivo effects of Platinum azidothymidine (Pt-AZT) upon HCC. To our knowledge, this is the first investigation to examine the in vivo effects of Pt-AZT on telomerase and Bcl-2 genes expression in rats with HCC that can demonstrate its potential as a new drug for HCC treatment. Also, in this study, the association between the expressions of these two genes in HCC was examined.
Materials and Methods :
Animal grouping and cancer induction in rats: Pathogen-free male Wistar rats whose average weights were about 50 gr were purchased from Razi Institute of Karaj in Iran and were maintained under standard conditions for two weeks for acclimatization. The animals had free access to industrialized food and water.
To study the effects of Pt-AZT on HCC and compare its effects with AZT (Sigma, Munich, Germany), four groups of pathogen-free male Wistar rats (n=100) were included in the study. Group A containing 25 healthy rats was considered as the control group. Preneoplastic lesions were induced in the liver of all remaining animals (n=75) using Solt-Farber resistant hepatocyte (RH) protocol as reported previously 31. After approving the cancer induction by a pathologist, they were randomly divide into three groups i.e. B, C and D (n=25 in each group).
Two weeks after the beginning of cancer induction in the rats, groups B, C and D received 200 mg/kg body weight (BW) of diethyl nitrosamine by IP injection for the initiation phase of hepatocarcinogenesis. The details of induction method were according to the reported standard protocols as described previously 31.
Histopathological studies: After preparation of paraffin-embedded blocks from samples, the slides were prepared and using Hematoxylin & Eosin (H&E) procedure, they were reviewed by a pathologist for confirming preneoplastic lesions in the studied animals 31. Drug treatments and biochemical studies were started after confirming preneoplastic lesions in the animals.
Drugs treatment: After confirmation of preneoplastic lesions on rat’s liver, groups C and D were treated by intraperitoneal injection (IP) of 0.9 mg/kg/day of Pt-AZT and 0.3 mg/kg/day of AZT, respectively 38 for 14 days. These drug concentrations were selected according to Jeng’s report 4 and our preliminary studies. At the end of the protocol, all rats were gradually sacrificed and molecular studies were carried out as follows.
RNA extraction: RNA was extracted from rat liver using commercially available kit (QIAzol Lysis Reagent) (Qiagen, Frankfurt, Germany). Slices of rat liver (50-100 mg) were prepared for extraction and all procedures were carried out according to the protocol of kit’s manufacturer.
cDNA synthesis of extracted RNA: cDNA was synthesized using commercially available kit (Transcriptor First Strand cDNA Synthesis Kit, Roche, Bavaria, Germany). The protocol was according to the kit’s manufacturer.
Quantitation of gene transcript by real-time quantitative RT-PCR: All real-time quantitative PCRs were performed by an ABI 7500 Real-Time PCR System (Applied Biosystems company, California, USA), utilizing Taqman reagents (Applied Biosystems company, California, USA) according to the manufacturer’s instructions. Amplification of PCR products was quantified during PCR by measuring fluorescence associated with binding of Taqman dye incorporated into the reaction mixture to double-stranded DNA.
The sequences of the oligonucleotide used in PCR-amplification of telomerase, B-cl2 and β-actin (a housekeeping gene as reference gene) are shown in table1. Briefly, each PCR test was performed in a 20 µl total volume mixture containing 10 µl of Taqman master mix Ex Taq (2x), 0.4 µl of ROX Reference Dye (50x), 1 µl of the mixture of Taqman probe and primer with 20x concentration and 2 µl of cDNA samples and then final volume reached to 20 µl with double distilled water (7 µl of dd water). After an initial denaturation step of 50°C for 2 min, an annealing step of 95°C for 10 min, elongation step of 58°C for 1 min, for both sense and antisense were followed in the reaction.
The Δct of target genes was obtained by subtracting the threshold cycle (ct) of target genes from those of β-actin as endogenous control and was calculated for each group separately. Then, by subtracting the Δct of target genes from Δct of control group, ΔΔct was calculated. The fold change of the target gene was estimated by using the following formula: fold change of target gene=2-ΔΔct.
Statistical analysis: The SPSS statistical software package version 16 was used for statistical analyses. A p-value<0.05 was considered significant. A nonparametric independent sample Mann-Whitney U-test was used to compare Bcl-2 and telomerase expression between related studied groups.
Results :
Histopatological findings: There were no preneoplstic lesions in normal rats in the control group at any level of our study, also there were not any abnormal preneoplastic lesions on hepatocyte of normal rat after treatment with AZT or Pt-AZT (Figure 1), but there were some necrosis and hemorrhage after treatments with these drugs (Figures 1G and H).
Preneoplstic lesions in rat liver lobes were developed following the neoplasm induction in untreated and medicine-treated groups. At the end of preneoplastic lesion induction protocol, there were few numbers of lesions and the sizes of them were small and with passage of time both size and number of lesions increased. After drug treatments, sizes of some lesions decreased and some small lesions disappeared; but the efficacy of Pt-AZT expression was higher compared to those of AZT.
According to pathological reports, there were no metastases after ending of (8 weeks) Solt Farber protocol in other organs such as stomach and lungs and because of small preneoplastic lesions after 8 weeks there were no significant changes in weight of rat liver.
Gene expression results: The Bcl-2 and telomerase expression in studied groups are shown in figure 2. Comparing group C with group B, there was a significant decrease in Bcl-2 expression (0.41±0.276 vs. 7.25±11.6, p<0.001) and telomerase expression (0.473±0.231 vs. 5.137±5.08, p<0.001) in group C. Also comparison between C and D groups showed lower expressions of Bcl-2 (0.41±0.276 vs. 4.93±5.18, p<0.001) and telomerase (0.473±0.231 vs. 3.48±4.02, p<0.001) in group C. These results indicated lower expression of these two genes in Pt-AZT treated group compared to AZT treated group. Expression of Bcl-2 in group B was higher as compared with group A (7.25±11.6 vs. 0.328±0.23 p<0.001). Similar results were found for telomerase (5.137±5.08 vs. 0.37±0.252, p<0.001) showing higher expression in group B. There was also higher expression of these two genes in group D as compared with group A. Bcl-2 and telomerase expressions were not significantly different between D and B groups. Similar results were obtained in comparing A and C groups.
Our results indicated a significant direct correlation between telomerase and Bcl-2 expression in untreated rats (r=0.54, p=0.006), however, there was no significant correlation between telomerase and Bcl-2 expression in other studied groups.
Discussion :
Our study confirmed the inhibitory effects of Pt-AZT on HCC and allows us to study anticancer effect of this new synthetic compound in vivo. In this study, it was found that Pt-AZT is more effective than AZT in HCC inhibition in the rats that cancer was induced by resistant hepatocyte Solt-Farber protocol. Our findings showed that telomerase and Bcl-2 expression in Pt-AZT-treated rats was lower than those of AZT-treated ones (Figure 2). Also similarly, these factors were reduced in AZT-treated rats compared to untreated HCC group (Figure 2). A study reported that AZT blocks telomerase expression and activity which effectively inhibits tumor growth and liver metastasis induced by the carcinogen diethyl-nitrosamine (DEN) in rats 3. Our finding indicted that telomerase expression increased after HCC development in untreated group but in the C and D groups that were treated with Pt-AZT and AZT, respectively it significantly decreased. These changes in telomerase and Bcl-2 expression can be novel tumor biomarkers to detect HCC either at primary or progressive stage. Our finding indicated that Pt-AZT effectively inhibited telomerase and Bcl-2 whose expressions significantly increased in untreated HCC group.
As mentioned earlier, PT-AZT was more effective than AZT in inhibition of telomerase and Bcl-2 expression and then metastasis and progression of HCC because of platinum atom in its molecular structure. Some reports confirming our results are mentioned in the following paragraphs.
Some authors reported that platinum derivatives of some drugs had more inhibitory effects on cancers than original drugs (without platinum) 32-35. Coluccia et al reported that platinum compound of acyclovir (antiviral drug) had anticancer properties, and had broader and stronger effect than acyclovir and had the same effect as cisplatin among anticancer properties 32. Muscella et al reported that a new platinum called Pt-ACD [Pt (O, O′-acac) (γ-acac) (DMS)] could induce apoptosis in various human cancer cells including human breast carcinoma cells (MCF-7) that are resistant to cisplatinum 33. Abu-Surrah et al explained the molecular structure of a vast majority of platinum compound groups and emphasized their powerful anticancer effects 34. Ruiz et al reported that a new platinum and palladium complex has anticancer effects about 8-fold more active than cisplatin against the human breast cancer cell line T47D and indicated this effect is due to the presence of platinum or palladium atoms in their molecular structure 35. All the above reports indicated that platinum element in structure of some anticancer drug will give them higher anticancer properties.
Also in addition to platinum atoms in the molecular structure of Pt-AZT which showed anticancer properties as previously mentioned, AZT component in Pt-AZT showing anticancer effects decreased expression of the two mentioned genes. Some studies confirmed our results as follows.
Jeng et al reported that AZT blocks telomerase and effectively inhibits tumor growth and liver metastasis induced by the carcinogen diethylnitrosamine (DEN) in rats 3. Chen et al showed synergistic effect of AZT with arsenic oxide in treatment of HCC with decreased expression of telomerase and increased levels of caspase 3, 36. Slamenova et al reported that AZT was responsible for widely destroying DNA strands in cancer cells 37. AZT also can inhibit telomerase activity and induced apoptosis in cultured-cancer cells 38. Our results emphasized that telomerase may be an important factor in cell growth and therapy. Tejera et al using mouse mammary carcinoma cell culture showed AZT has anti-metastatic potential and can reduce telomerase expression 25.
Some reports showed how AZT exerts its anticancer effects on cancer cells. AZT is probably phosphorylated intracellularly and then attaches to telomere region of DNA that can cause shortening and disruption of this region due to induction of apoptosis 39. AZT can also lower telomerase concentration and thereby shorten the telomeres 23.
Also platinum component of Pt-AZT exerts its anticancer effects on cancer cells in the following way. Cancer cells by excessive proliferation of their genome and then excessive absorption Platinum compounds respect to normal cells due to creation adducts with base sequences especially with guanines of DNA, these adduct especially 1,3 intrastrand guanines which form 97% adducts in DNA not repaired and finally due to apoptosis of cancer cells 40.
The relationship between Bcl-2 concentration and resistance to drug treatment remains controversial. According to our results showing the lower Bcl-2 expression and then concentration in Pt-AZT- treated rats compared to AZT-treated group 41, there was less resistance to the drug in the former compound. Comparing the extension of apoptosis that was wider in Pt-AZT-treated group than AZT, greater inhibition of Bcl-2 gene expression and concentration decline by Pt-AZT in comparison to AZT was observed.
Some studies indicated inverse relationship between Bcl-2 expression and concentration and resistance to anticancer drugs 42,43. Beale et al showed a statistically significant inverse correlation between inhibition of cell line growth and Bcl-2 levels in human ovarian carcinoma cells treated with cisplatin; over-expression and therefore increased concentration of Bcl-2 in these cells led to resistance to cisplatin compared to the control 44. In this study, it is concluded this relationship depends on some factors such as nature of anticancer drug and the applied concentration.
Our results indicated a significant direct correlation between telomerase and Bcl-2 expression in untreated rats (r=0.54, p=0.006), but there was no significant correlation between telomerase and Bcl-2 expression in other studied groups. Furthermore, there is discrepancy in the relationship between telomerase and Bcl-2 expression in some reported studies 44-46. However, Iida et al reported a possible relationship between telomerase and Bcl-2 expression in colorectal carcinoma 45. Elkak et al reported no relationship between telomerase and Bcl-2 expression in human breast cancer 46. Also, Ohmura et al suggested that Bcl-2 expression and therefore concentration was conversely correlated with telomerase expression and activity (similar to our findings) and the biological role of Bcl-2 expression differs by degree of tumor aggressiveness in low grade tumor 47. Similar to the above mentioned study, induction of HCC in our studied animals was in primary level (preneoplastic lesions); therefore, the degree of tumor aggressiveness was in low grade and the inverse correlation between telomerase expression and Bcl-2 expression was statistically significant.
Conclusion :
In this study, by inducing HCC to animal model, the inhibitory effects of Pt-AZT in animal was investigated and it was concluded that Pt-AZT can reduce Bcl-2 and telomerase expression more effectively compared to AZT (the highest levels of Bcl-2 and telomerase expression were found in untreated HCC group). Our research was an in vivo study and hence it can be considered as an advantage of the study that it differs from most of the studies in this field. The novel finding in our study was introduction of Pt-AZT as a new anticancer drug in vivo that can be more efficient than AZT. However, further studies are needed to shed light on inhibitory effect of Pt-AZT on the growth of liver tumor in rats by extending apoptosis.
Acknowledgement :
This study was supported by a grant from Hamadan University of Medical Sciences, Hamadan, Iran. This is a part of A. Sabokrouh PhD thesis. The authors would like to thank Dr. Nahid Shahabadi for providing Pt-AZT drug.
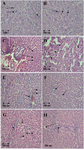
Figure 1. Cytological changes during preneoplastic lesion induction and after treatment with Pt-AZT and AT in rats’ liver (H&E staining method). A and B) rat liver with malignant cells. The arrows show the enlarged nucleuses of preneoplastic cells (original magnification 400× in A and 100× in B); C and D) rat’s liver after treatment with Pt- AZT. The arrows show disrupted nucleus (karyolysis) which is the sign of apoptosis in D slide, arrows also show in some parts necrosis and hemorrhage (original magnification 100×); E and F) rat’s liver treated with AZT. Arrows show karyolysis and pyknosis, also necrosis and hemorrhages are seen in some parts (original magnification 100×); G and H) normal rat’s liver treated with AZT and Pt-AZT, respectively. There were no preneoplastic lesions in some parts and there were some necrosis and hemorrhage (original magnification 100×).
|

Figure 2. Expression of Bcl-2 A) and telomerase B) in different experimental groups (mean±SEM). A) Control group, B) Untreated cancer group, C) Cancer group treated with Pt-AZT, D) Cancer group treated with AZT.
*p<0.001.
|
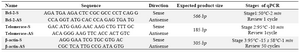
Table 1. Primers of target genes in sense and antisense with qPCR conditions
|
|