Variable Number Tandem Repeat (VNTR) Genotyping of Hydatidiform Mole in Iranian Patients
-
Pakzad, Zahra
-
Department of Medical Genetics, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran
-
 Mozdarani, Hossein
Department of Medical Genetics, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran, Tel: +98 21 82883830; Email: mozdarah@modares.ac.ir
Mozdarani, Hossein
Department of Medical Genetics, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran, Tel: +98 21 82883830; Email: mozdarah@modares.ac.ir
-
Department of Medical Genetics, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran
-
Izadi-Mood, Narges
-
Mirza Koochak Khan Hospital, Iran University of Medical Sciences, Tehran, Iran
-
Niromanesh, Shirin
-
Mirza Koochak Khan Hospital, Iran University of Medical Sciences, Tehran, Iran
Abstract: Background: Classification of molar gestation into Complete Hydatidiform Mole (CHM) and Partial Hydatidiform Mole (PHM) is done according to clinical, ultrasonographic, histologic and genetic criteria. However, making a distinction between CHM and PHM using histologic criteria alone may be difficult and several studies have shown that misclassifications are frequent, even for experienced pathologists. CHM is the most common precursor to choriocarcinoma and heterozygous moles carry an increased predisposition to transformation. Methods: Formalin-fixed, paraffin-embedded tissue sections of patients as well as peripheral blood of patients and their partners’ were collected in EDTA tubes. Tissue samples were obtained by curettage. Histological evaluation was performed on routine section stained with Hematoxylin and Eosin. Variable Number Tandem Repeats (VNTRs) genotyping was performed for 30 cases in two groups of CHM (n=21) and PHM (n=9), with Polymerase Chain Reaction (PCR) amplification of 2 different polymorphic loci, namely the Col2A1 and D1S80. Results: The results of DNA analysis by VNTR genotyping showed that in 16 cases of CHM, amplification of the VNTR polymorphic loci showed androgenetic mono-spermic moles (homozygote) and in 5 cases of CHM androgenetic dispermic moles (heterozygote) in molar tissue. In cases of PHM, 6 samples were triploid dispermic and 3 samples were diploid biparental. Conclusion: This study confirmed that VNTR genotyping can identify the parental source of polymorphic alleles in hydatidiform mole. Compared to STR genotyping, VNTR genotyping was performed by PCR amplification of several minisatellite markers of DNA. This method significantly requires less time and is cost-effective.
Introduction :
Hydatidiform Mole (HM) (OMIM 231090) is an abnormal gestation characterized by significant hydropic enlargement and variable trophoblastic proliferation involving part of or all chorionic villi. HMs are sub-classified into Complete HM (CHM) and Partial HM (PHM), based on clinical features, histopathology and genetic differences 1-3.
HMs are relatively common and occur in approximately 1 in 1500 pregnancies in Europe and North America. This incidence varies between ethnic groups and is 2-10 times higher in some countries of Latin America, the Middle East, and the Far East with the highest frequencies being in Mexico, Iran and Indonesia 4-7.
Karyotype and genotype analyses have shown that sporadic moles may have different genotypic types with the majority of the CHMs being diploid androgenetic and the majority of the PHMs being triploid diandric dispermic. Among androgenetic moles, the majority are mono-spermic (homozygous) and 10-20% are dispermic (heterozygous) 8-12. Studies have suggested that heterozygous mole may have a more malignant potential than its homozygous counterpart 13,14.
With the increased use of high resolution ultrasonography in early pregnancy, the diagnosis and sub-classification of hydatidiform moles have become more difficult, as CHMs are now commonly evacuated at an earlier stage and before the development of classical morphologic and histologic features. Recently, pathologists have relied on molecular techniques, such as DNA flow cytometry, chromosome in situ hybridization, polymerase chain reaction-based genotyping or HLA typing, showing DNA content differences for CHM and PHM 15-17. Approximately 75% of clinically ascertained molar pregnancy are CHMs and mostly of diploid, androgenetic origin, and the remaining 25% are PHMs and mostly of diandric, triploid origin 18,19. Vanda Repiska et al suggested that PCR amplification of polymorphic VNTR regions will be a useful tool in helping to determine relative risk of choriocarcinoma in patients with complete hydatidiform moles. Closer clinical surveillance of patients with heterozygous moles may prove to be an important part of management in the future 20.
Persistent gestational trophoblastic disease develops after a CHM in 10 to 30% of cases and after a PHM in 0.5 to 5%. Choriocarcinoma arises in up to 3% of CHM and is rare but reported to be one of the consequences of PHM 21,22. Despite the difference in risk for persistent disease or metastases, the post evacuation surveillance recommendations are identical with frequent monitoring of serum human chorionic gonadotropin levels until normalization and monthly monitoring for 6 month thereafter 23,24.
Each human somatic cell contains 6.4 billion base pairs of DNA. Portions of this DNA encode over 30,000 genes, while the remainders are non-coding. The human genome contains hundreds of highly polymorphic segments, each of which is characterized by allelic variation and attributed to the difference in the copy number of the tandemly repeated DNA sequences, such as Variable Number of Tandem Repeats (VNTRs). There are two principal families of VNTRs: minisatellite and microsatellite. Minisatellites are composed of different repeated "core" DNA sequences arranged in tandem. The size of the core sequence can vary from 7 to 100 bp in different VNTRs. Due to their high polymorphic content, VNTR is a useful tool in linkage analysis, forensic identification, paternity testing and population genetic studies 25-27. VNTRs or minisatellite loci are polymorphic and due to the variation in the number of copies of their repeat unit, VNTRs are multiallelic, so they display variation in length of the repeats and don٫t vary from one generation to the next. They are easily amenable to Polymerase Chain Reaction (PCR). Regarding the above mentioned criteria, these markers would be useful and informative in studying genetic variation among human population 28. In a study performed in Iran, it was shown that both ApoB and D1S80 are very polymorphic in Iranian populations 26. The Col2A1 minisatellite as one of VNTR markers, located at 3' of last exon of Col2A1 gene has been shown to be highly variable and informative 29.
Therefore, the aim of this study was to show the value of VNTR amplification for genotyping of CHM and PHM.
Materials and Methods :
Patients: Thirty cases were selected from gynecologic oncology service. Formalin-fixed, paraffin-embedded tissue sections of the patients were collected from the routine gynecologic pathology service files of the Mirza Koochak Khan Hospital, a referral gynecologic hospital in Tehran, Iran. Before sample collection, the study was reviewed and approved by the Tarbiat Modares University Ethics Committee.
The mean age of patients with complete mole was 26.4 years (range, 15-41 years) and in patients with partial mole, it was 30 years (range, 24-37 years). Peripheral blood of patients and their partners’ were collected in EDTA tubes. Tissue samples were obtained by curettage. Histological evaluation was performed on routine section stained with Hematoxylin and Eosin and diagnoses were made by gynecologic pathologist.
For each specimen, the maternal deciduas and molar villi on paraffin block were separated using scalpel and a consecutive section stained with Hematoxyline and Eosin (Figure 1). Then three, 20 µm thick sections from FFPE tissue blocks were collected in 1.5 micro tubes for each case.
DNA isolation: DNA was isolated from FFPE tissue as well as from peripheral blood of patients and their partners. DNA extraction from peripheral blood was performed using modified salting out method. For each extraction, 500 µl blood (EDTA, whole blood) was used. Then, 1000 µl cell lysis buffer was added into the sterile 1.5 ml tube and centrifuged at 7000 RPM for 4 min and the supernatant was discarded. This step was repeated once more. To the remaining white pellet, 300 µl nuclei lysis buffer was added and incubated at RT for 30 min until the pellet was re-suspended. Then, 100 µl NaCl (Merck, Germany) 6M and 600 µl choloroform (Merck, Germany) were added and centrifuged at 7000 RPM for 4 min. The supernatant (upper layer) was transferred to a new 1.5 tube and 1000 µl cold Ethanol (Merck, Germany) 100% (-20°C) was added. The tube was inverted 5-6 times gently until Hank of DNA was obtained, then centrifuged at 1000 RPM for 2 min. The supernatant was discarded and cold Ethanol (Merck, Germany) 70% (-20°C) was added, again tube was centrifuged. The supernatant was discarded and 100 µl distilled water was added to re-suspended DNA.
DNA was prepared from tissue using modified Phenol-chloroform method. In each case, three, 20 µm-thick sections from FFPE tissue blocks were collected in 1.5 micro tubes. Briefly, the tissue sections were deparaffinized with Xylene (Merck, Germany), washed with 100%, 96% and 70% Ethanol (Merck, Germany) and dried in thermo block. The dried tissue was then incubated in 200 µl tissue lysis buffer (Genetbio, Korea) and 50 µl proteinase K (Genetbio, Korea). After overnight incubation at 46°C, the tube was incubated at 75°C for 1 hr until complete digestion of tissue. Then, 250 µl Phenol-Choloroform-Isoamyl alcohol (Sinaclon, Iran) was added and centrifuged. The upper layer was transferred to a new 1.5 tube and equal volume of Isopropanol alcohol (Merck, Germany) was added and then incubated at 4°C overnight. The next day, the tube was inverted several times and then centrifuged at 13000 RPM for 30 min. The supernatant was discarded and 50 µl distilled water was added to eluted DNA.
PCR amplification: The DNA samples were subjected to polymerase chain reaction (PCR) amplification of 2 VNTRs (variable number tandem repeats) polymorphic loci, namely the Col2A and D1S80. The primer sequences for 2VNTR locus 29-31 are given in table 1.
All the DNA samples were subjected to PCR with slight modification in a thermocycler (CreaCon- Netherlands). To determine the genetic origin of each HM, 1 µl DNA from the patient and her partner and 1.5 µl DNA from each of the HM were amplified using two pairs of primers (BioNEER, South Korea) in a final reaction volume of 25 µl containing DNA, 2 Taq Master mix (Amplicon, Denmark) 12.5 µl and 50 pmols of each primer.
The cycle conditions were as follows: for Col2A1, 30 cycles of denaturation at 94°C for 30 s; annealing at 60°C for 50 s; extension at 72°C for 50 s, for D1S80, 30 cycles of denaturation at 94°C for 30 s; annealing at 70°C for 50 s; extension at 72°C for 50 s. The initial denaturation and final extension were carried out at 94°C for 5 min and 72°C for 10 min for Col2A1 and D1S80.
Following amplification, 8 µl of each PCR reaction product was analyzed by electrophoresis in a 2% agarose gel with a 100 bp molecular weight marker, stained with ethidium bromide and documented in a gel documentation system. In the interpretation of genotyping results, the allele sizes of minisatellite polymorphisms found in the molar villi were then compared with the corresponding allele sizes observed in the maternal and paternal samples as shown in figures 2-4.
Results :
The results of DNA analysis by VNTR genotyping as well as other related information about patients including clinical parameter and histologic diagnosis are shown in table 2. In 16 cases of CHM, amplification of the VNTR polymorphic loci showed androgenetic mono-spermic moles (homozygote) in molar tissue (76.2%). In 5 cases of CHM, androgenetic dispermic moles (heterozygote) were seen in molar tissue (23.8%). In cases of PHM, 6 samples were triploid dispermic (66.7%) and 3 samples were diploid biparental (33.3%).
Discussion :
CHMs are generally diploid and androgenetic in origin, all 46 chromosomes being derived from the father. They may be monospermic, arising by fertilization of an enucleate egg by a single spermatozoon which then doubles to provide a diploid chromosome complement, or dispermic arising from fertilization of an enucleate egg by two spermatozoa. PHMs are usually triploid, the extra haploid set of chromosome can have either a maternal origin (digynic triploidy) or a paternal one (diandric triploidy) 14,15.
Our results showed that 76.2% cases of CHM were homozygous and 23.8% heterozygous. Also, in cases of PHM, 66.7% were triploid dispermic and 33.3% diploid biparental. These results are consistent with reports in other investigations 8,11.
Persistent gestational trophoblastic disease develops after a CHM in 10 to 30% of cases and after a PHM in 0.5 to 5% 21,22. Homozygous and heterozygous CHM are two genetically distinct entities which can only be distinguished on the basis of genetic analysis and heterozygous mole may have a more malignant potential than its homozygous counterpart 16,17. In this study, five of 21 CHM cases, analyzed by genotyping, were found to be heterozygous and the remaining cases were homozygous.
Although many cases of hydropic abortion can be diagnosed on the basis of histology alone, it is difficult to estimate the proportion of cases evacuated at an early gestational age 1,2,15. Thus, accurate diagnosis of abnormal product of conception specimens and subsequent classification as complete or partial mole is mandatory to ascertain the actual risk of persistent GTD and to determine the appropriate nature and duration of clinical follow-up care. More advanced molecular genetics techniques have subsequently been applied to resolve this differential diagnosis problem 15-17.
The PCR amplification of polymorphic VNTR region will be a useful tool in helping to determine relative risk of choriocarcinoma in patients with complete hydatidiform moles 20. VNTRs are the most informative markers for genetic studies. Although each VNTR locus is usually associated with a large number of different alleles in any population, each individual carries two alleles, one on each of the two homologous chromosomes. The major methodological advantage of VNTR is that all alleles are stable across generation, they are inherited in a Mendelian fashion and do not vary from parents to offspring 25,27.
Other techniques that can be used for genetic evaluation of molar pregnancy include Fluorescence In Situ Hybridization (FISH), flow cytometry and fluorescent microsatellite genotyping. However, comparison of VNTR genotyping with other methods showed that VNTR method is very simple, thus does not require special device and also it is less time-consuming and more cost-effective.
In this study, it was shown that VNTR genotyping can identify the parental source of polymorphic alleles and thus discern androgenetic diploidy (homozygous and heterozygous), diandric triploidy, and biparental diploidy, which allows for specific diagnosis of CHMs, PHMs, and NMs, respectively. In a similar study, choriocarcinoma analyzed by PCR amplification of polymorphic VNTR in 9 out of 10 cases was identified as herterozygous 20.
The reliability of the diagnosis is crucial for appropriate counseling and to determine if a patient falls in to a "short-term" or "long-term" follow-up. In some instances, for example in women having a "lesion suspicious for HM", the diagnosis could be confirmed or ruled out, thus avoiding an unnecessary follow-up. This may be of particular importance in "older patients" having difficulties in conceiving and for whom a one-year wait may be extremely distressing 9,13,16,24,32.
Conclusion :
In conclusion, the majority of cases of hydropic abortion can be diagnosed based on histological feature alone, but molecular biological techniques will be necessary to evaluate the proportion of cases evacuated at an early gestational age. Our results indicate that VNTR genotyping, a relatively simple, reliable, cost-efficient and rapid procedure, can be used successfully for diagnosis of HM and distinguishing CHM from its mimics, which is clinically important because the majority of persistent trophoblastic diseases and choriocarcinomas arise from CHM.
Acknowledgement :
This work was supported by Research Department of the Faculty of Medical Sciences, Tarbiat Modares University. The authors would sincerely thank for contribution of all participating individuals, especially patients in this study.
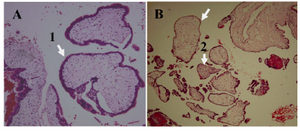
Figure 1. H&E staining of hydatidiform mole. A) CHM characterized by, 1: hydropic villi with circumferential trophoblastic hyperplasia ×100 B) PHM characterized by, 1: hydro-pic and 2: fibrotic villi with focal mild trophoblastic hyperplasia ×100
|
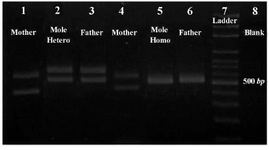
Figure 2. Results of the PCR amplification of D1S80 VNTR locus, line 1: mother; line 2: CHM of heterozygous; line 3: father; line 4: mother; line 5: CHM of homozygous; line 6: father; line 7: ladder; line 8: blank
|
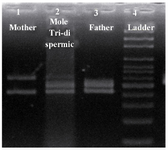
Figure 3. Results of the PCR amplification of D1S80 VNTR locus, line 1: mother; line 2: PHM of triploid dispermic; line 3: father; line 4: ladder
|
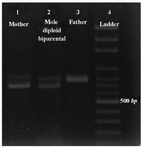
Figure 4. Results of the PCR amplification of Col2A1 VNTR locus, line 1: mother; line 2: PHM of diploid biparental; line 3: father; line 4: ladder
|

Table 1. Minisatellite primers
|
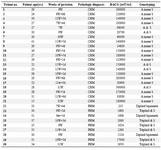
Table 2. Clinical parameter, histological diagnosis and genotyping findings of patients examined
|
|