Comparative Proteomics Study of Streptozotocin-induced Diabetic Nephropathy in Rats’ Kidneys Transfected with Adenovirus-mediated Fibromodulin Gene
-
Maleki, Akram
-
Zanjan Metabolic Disease Research Center, Zanjan University of Medical Sciences, Zanjan, Iran
-
 Ramazani, Ali
Zanjan Pharmaceutical Biotechnology Research Center, Zanjan University of Medical Sciences, Zanjan, Iran, Tel: +98 241 4273636; Email: ramazania@zums.ac.ir
Ramazani, Ali
Zanjan Pharmaceutical Biotechnology Research Center, Zanjan University of Medical Sciences, Zanjan, Iran, Tel: +98 241 4273636; Email: ramazania@zums.ac.ir
-
Zanjan Pharmaceutical Biotechnology Research Center, Zanjan University of Medical Sciences, Zanjan, Iran
-
Foroutan, Maryam
-
Department of Molecular Medicine & Genetics, School of Medicine, Zanjan University of Medical Sciences, Zanjan, Iran
-
Biglari, Alireza
-
Department of Molecular Medicine & Genetics, School of Medicine, Zanjan University of Medical Sciences, Zanjan, Iran
-
Ranjzad, Parisa
-
Vascular Gene Therapy Unit, Research School of Clinical & Laboratory Sciences, Manchester Academic Health Science Centre, The University of Manchester, Manchester, UK
-
Awsat Mellati, Ali
-
Zanjan Metabolic Disease Research Center, Zanjan University of Medical Sciences, Zanjan, Iran
Abstract: Background: Transforming Growth Factor-beta (TGF-β) activation appears to be crucial for tissue injury in Diabetic Nephropathy (DN). Fibromodulin, the small leucine-rich proteoglycan, has been proposed to be the potent TGF-β modulator. In this study, the therapeutic effects of fibromodulin in the kidneys of streptozotocin (STZ)-induced diabetic rats were investigated.
Methods: Diabetic rats received intraperitoneal (IP) injections of recombinant adenovirus expression vectors (RAd5) containing fibromodulin (RAd-FMOD) and were killed after 10 weeks. Proteins were isolated from the rat kidney and separated using two-dimensional gel electrophoresis. The differentially expressed proteins were analyzed using Matrix-assisted laser desorption/ ionization time-of-flight mass spectrometry (MALDI-TOF-MS).
Results: Ten spots were identified using MALDI-TOF-MS. The identified proteins were primarily responsible for cell metabolism, cytoskeleton formation, and oxidative stress. RAd-FMOD treatment markedly attenuated the albuminuria in diabetic rats.
Conclusion: Taken together, these results provide a valuable clue in exploring the mechanism underlying the therapeutic effects of fibromodulin in diabetic nephropathy suggesting that it can be a potential agent in the treatment of this disease.
Introduction :
Diabetic Nephropathy (DN), also known as Kimmelstiel-Wilson syndrome, is a progressive kidney disease caused by angiopathy of capillaries in the kidney glomeruli. It is characterized by nephrotic syndrome and diffuse glomerulosclerosis and accounts for approximately 50% of end-stage renal failure cases.
Although there have been significant breakthroughs in the last decade with regard to the prevention and treatment of diabetic kidney disease 1, the pathophysiological mechanisms of DN remain poorly understood. Research provides ample evidence that Small Leucine-Rich Proteoglycans (SLRPs) such as decorin, biglycan, fibromodulin, and lumican in the extracellular matrix are deeply involved in the regulation of inflammatory and fibrotic renal disorders and are the binding partners of TGF-β 2,3. Fibromodulin is a SLRP that along with other SLRPs such as decorin can interact with several different cell surface receptors, cytokines, ECM components, and growth factors such as TGF-β ligands to modulate their activity 4. Fibromodulin possesses a central region containing leucine-rich repeats with 4 keratan sulfate chains, flanked by terminal domains containing disulphide bonds. Owing to the interaction with type I and type II collagen fibrils and in vitro inhibition of fibrillogenesis, the encoded protein may play a role in the assembly of extracellular matrix. It may also regulate TGF-β activities by sequestering TGF-β into the extracellular matrix. Numerous evidences support the role of the TGF-β and downstream Smad signaling in the development and progression of renal fibrosis. TGF-β activation appears to be crucial for tissue injury in DN. Over-expression of TGF-β cytokine has been identified as the key mediator of chronic progressive kidney fibrosis, thereby playing an important role in the pathogenesis of this disease 5. The profibrotic actions of TGF-β make it an ideal therapeutic target for renoprotective agents.
Fibromodulin has not been studied in association with DN; therefore, this study aimed to investigate the effects of fibromodulin in DN treatment. Here we assessed the effects of fibromodulin in DN streptozotocin (STZ) induced rats by proteomics approach.
Materials and Methods :
Recombinant adenovirus construct: Recombinant adenovirus expression vectors (RAd5) containing fibromodulin (RAd-FMOD) or lacZ (RAd-lacZ) cDNA were gifted by Paul Kingston, Gene Therapy Unit, University of Manchester. RAd vectors were E1/E3-deleted first-generation adenoviruses, in which the cassette containing a recombinant transgene and promoter were inserted in place of the E1 region. lacZ and FMOD expression was driven by a murine CMV (mCMV) promoter. The Woodchuck hepatitis virus Post-transcriptional Regulatory Element (WPRE) resulted in significantly higher lacZ expression levels (8-fold) in primary endothelial cells 6. The efficiency of these vectors were confirmed in a previous study 7.
Bioactivity of RAds encoding fibromodulin in vitro: HEK 293 cells were obtained from Pasteur Institute of Iran, maintained in DMEM, supplemented with 10% fetal bovine serum, and grown in 5% CO2 at 37°C. Confluent HEK cells (2×105 cells/60 mm dish) in conditioned 2% medium were treated with RAd-FMOD (MOI=1000) or RAd-lacZ. After 5 hr incubation, the supernatant was discarded and 5 ml of the standard medium was added in every 60 mm dish. Uninfected cells were cultured in the same conditions served as negative control. The cells were collected at 72 hr to detect FMOD mRNA using Reverse Transcriptase Polymerase Chain Reaction (RT-PCR).
Animal mode: Adult male Sprague-Dawley rats with a body weight ranging from 250 to 300 g were used. After 24 hr fasting, the rats were injected with a single IP injection of 50 mg/kg STZ (Sigma, USA). Three days (72 hr) after STZ injection, blood glucose levels were measured via the tail vein using a glucometer (Accu-Chek Active). The criterion of high blood glucose levels (250 mg/dl) in diabetes was considered. At 8 weeks, 24 rats were randomly divided into 4 groups (n=6). One group (STZ-fibromodulin group) received a 0.6 ml (1×109 PFU/ml) IP injection of RAd-fibromodulin. Another group (STZ-lacZ group) received a 0.6 ml (1×109 PFU/ml) IP injection of recombinant adenovirus encoding lacZ. The third group (STZ-PBS group) received a 0.6 ml IP injection of PBS. At this time, the vehicle control rats (PBS control) that were not induced with diabetes also received a 0.6 ml IP injection of PBS.
At 10 weeks after STZ injection, individual rats were placed in metabolic cages to obtain 24 hr urine collections. The animals were then sacrificed, and blood and kidneys were collected for analysis. The urine samples were kept at -20°C until later measurements of creatinine (Jaffé assay) and albumin (Assaypro Co.). Kidneys were quickly removed and thoroughly washed with PBS to remove blood and kept at -80°C for proteome and FMOD mRNA analysis. Kidney Hypertrophy Index (KHI) was calculated as the ratio of kidney weight (g) to body weight (kg). Urinary Albumin Excretion (UAE) values were calculated as the ratio of urinary albumin to creatinine (mg/g).
Sample preparation for two-dimensional gel electrophoresis (2DE): The frozen kidneys were cut into small pieces, washed with PBS, and ground into fine powder in liquid nitrogen with a pestle. The powder was then suspended in lysis buffer (CHAPS 4%, urea 6 M, thiourea 2 M, DTT 50 mM, Tris HCL 50 mM, pH=7.4) with protease inhibitor cocktail (Sigma) and 10 mM PMSF (Sigma) added in the ratio of 1:50 and 1:20 (v:v), respectively. The suspension was sonicated (UP200H, Hielscher) on ice for 3 cycles of 60 kHz, each lasting 30 s, incubated for 1 hr at room temperature, and centrifuged (14000×g at 4°C for 15 min). After 1 hr of incubation at room temperature, the homogenate was centrifuged (3-30 K, Sigma) at 20,000×g at 4°C for 30 min 8. Protein concentration was measured using the modified Bradford assay with BSA standards 9.
2DE: A fixed amount of 1000 μg of kidney proteins was taken from each sample and mixed with rehydration buffer (8 M urea, 4% CHAPS, 50 mM DTT, 0.2% Ampholyt, pH=3-10, 40×Fluka) to a volume of 250 μl, which was then loaded onto 13 cm IPG strips (GE Healthcare; linear pH=3-10). IPG strips were rehydrated for 17 hr at room temperature. 1D IEF was performed at 20°C by using the following parameters: 500 Vh, 800 Vh, 11,300 Vh, and 2,900 Vh for 15,500 Vh (protocol provided by the manufacturer; GE Healthcare, Uppsala, Sweden). The strips were first equilibrated for 15 min in an aqueous solution (pH=8.8) containing 50 mM Tris, 6 M urea, 30% (w/w) glycerol, 2% (w/w) SDS, and 1% DTT then for an additional 15 min in a solution (pH=8.8) containing 50 mM Tris, 6 M urea, 30% (w/w) glycerol, 2% SDS, 4% iodoacetamide, and trace amount of bromophenol blue. Second dimension SDS PAGE was performed on the SE 600 Ruby system (GE Healthcare) by using a homemade 12% vertical SDS-PAGE slab gels (140 mm×160 mm×1 mm). Second dimension SDS- PAGE was first run at a current of 15 mA/gel for 1 hr and then at a constant current of 30 mA/gel at 10°C until bromophenol blue reached the gel bottom 10. The resolved 2D spots were fixed in fixing solution (10% acetic acid, 40% ethanol, 50% deionized water) and stained with colloidal coomassie brilliant blue G-250 (Merck, Germany) 10. Differential analysis was performed using ImageMaster 2D Platinum 6.0 software tools (GE Healthcare) on a set of 3 independent gels per experimental condition. Statistical analysis of protein expression changes more than two-fold was performed in 2D gels prepared from 3 replicates in each group by using the student’s t-test based on volume % of matched spots, and statistical significance was assumed for p-values less than 0.05.
In-gel trypsin digestion, MS analysis, and database searching: The protein spots of interest were manually excised from preparative 2-D gels. In-gel tryptic digestion was performed after reduction with DTE and S-carbamidomethylation with iodoacetamide. Proteins were identified by proteomics service of University of York, York, UK. Tandem mass spectral data were submitted to database searching using a locally-running copy of the Mascot program (Matrix Science Ltd., version 2.1), through the Bruker BioTools interface (version 3.2).
Results :
Fibromodulin expression by adenovirus vector in cultured cells and kidneys: RT-PCR was performed using the mRNA which was extracted 72 hr after cell infection with RAd-FMOD to verify the expression of FMOD mRNA in HEK 293 cells. The mRNA signal for FMOD was detected in RAd-FMOD-infected HEK 293 cells but not in RAd-lacZ-infected cells or uninfected cells (Figure 1). The FMOD mRNA signal was also detected in RAd-FMOD-infected rats but not in RAd-lacZ rats or uninfected rats (Figure 1).
Effect of RAd-FMOD on KHI and UAE: As expected, the KHI measures in all diabetic control groups, including RAd-lacZ group were higher than those in healthy control group. The KHI of the RAd-FMOD group was slightly lower than that of the diabetic control group. However, there were no significant differences in KHI measures among all diabetic rat groups (RAd-FMOD, RAd-lacZ, and diabetic control; Figure 2A). Albuminuria was an early hallmark of DN. UAE measures in the diabetic control group was significantly larger than that in the PBS control group. UAE measures in the RAd-FMOD group were significantly smaller than those of the diabetic control group. By 10 weeks, UAE of the RAd-FMOD group decreased to 0.28 g/mg (p0.05 when compared with the RAd-lacZ group), which was similar to the value in PBS control group at this time point. Moreover, there was significant difference between UAE measures of the RAd-FMOD group and that of the RAd-lacZ group (Figure 2B).
2DE analysis results: Analyses of 2DE gel patterns between different groups provided the first clues to distinct alterations in the rat kidney proteome in the studied groups. Spots with more than two-fold changes in intensity compared with untreated controls in all the 3 replicate samples were identified and marked. Proteins showing altered levels of expression after RAd-FMOD treatments were identified using MALDI-TOF-MS. Of the spots selected for analyses (over 11 spots), successful identification was achieved for 10 of the proteins (Table 1).
Of the 10 proteins that were identified, 2 were up-regulated and 3 were down-regulated in the RAd-FMOD-treated group in comparison with diabetic kidney group. Moreover, 3 proteins were up-regulated and 4 were down-regulated in the RAd-FMOD-treated group in comparison with normal group. 1 spot appeared as de novo, and the other identified spot disappeared in RAd-FMOD-treated group and 5 proteins were up-regulated and 1 was down-regulated in the diabetic kidney group in comparison with normal group.
Discussion :
In this study, 10 out of 11 protein spots showing significant differences were identified using MALDI-TOF-MS. The results of this study showed that 4 out of 10 proteins (peroxiredoxin 6, cofilin 1, TGFβ1, and tubulin α) showed decreased expression and heat shock protein 60 (HSP60) showed increased expression in the RAd-FMOD-treated rats as compared with DN rats. Isocitrate dehydrogenase (ICDH) as a novel protein and albumin were identified as disappeared proteins in the RAd-FMOD-treated rats.
In this study, among 10 protein spots, regucalcin, HSP60, ICDH, cofilin 1, peroxiredoxin, tubulin α, and TGF-β1 which are known for their role in DN, were discussed.
Regucalcin is a calcium ion-binding protein that was significantly reduced in both diabetic and treated groups in comparison with the control group (2.8-fold decrease in the diabetic group and 8.5-fold in the treated group). It inhibits tRNA synthetase and plays a limiting role in protein translation. It activates cysteine protease, thus playing an important role in the turnover of proteins and affects superoxide dismutase activity in liver and kidney cells. The mRNA levels of this protein were reduced by 70% in the overnight fasting group as compared with that in the fed group 11.
It has been shown that liver regucalcin was reduced by 50% after 1-3 weeks of subcutaneous STZ injections, suggesting that it plays a key role in metabolic disorders such as diabetes 12. Because of the significant reduction of regucalcin in the treated group (8.5-fold) as compared with that in the diabetic group (2.5-fold), we concluded that fibromodulin probably plays a role in reducing regucalcin expression.
NADP-dependent ICDH was identified as a novel protein in the treated group. This enzyme plays an important role in response to oxidative stress. One of the fundamental roles of NADP+-dependent ICDH is the control of reduction potential in mitochondria and cytosol and defense against oxidative damage because this enzyme provides the required NADPH for antioxidant systems. Increase in glucose results in changes in ICDH in the glycosylation reaction, leading to a disruption in the integrity of the body's antioxidant defense by reducing the production of NADPH as an essential cofactor in the GSH cycle (glutathione recovery form), which may lead to pathological changes associated with ROS production 11. These results together indicate that up-regulation of ICDH in response to hyperglycemia may play an essential role in preventing the progression of DN, which is accompanied by ROS-induced cellular damage and fibrosis by providing NADPH, the reducing equivalent required for recycling reduced glutathione and low molecular weight antioxidant thiol proteins 13.
HSP60 protein showed increased expression in the treated group (2.8 times higher expression than that in the normal group and 1.8 times higher expression than that in the diabetic group). While association between altered chaperone function and development of various diseases has been shown, there is evidence that increased expression of HSP60 in type II diabetic patients is associated with nephropathy. One way of protecting the body against oxidative stress is induction of protective genes such as HSP family. The importance of these proteins is the response to oxidative stress in nephropathic patients 14,15. Considering the role of fibromodulin in reducing TGF-β, it can be expected that it also reduces the expression of HSP60. However, according to the results of this study, HSP60 expression showed an increasing level among groups treated with fibromodulin. Both hyperglycemia and glomerular hypertension are crucial determinants in the pathogenesis of DN and impose cellular stresses on renal target cells 16. The results of this study suggest that probably fibromodulin increased the expression of HSP60 through other mechanisms to cope with oxidative stress.
Peroxiredoxin 6: The expression of this protein was significantly increased in the treated group as compared with that in the diabetic and control groups. Peroxiredoxin is a protein that is involved in oxidative stress, and the relationship between protein oxidation and oxidative damage is known. It has been shown that the enzymes responsible for ROS in the cytosol (catalase; peroxiredoxin 6, 4, and 1; superoxide dismutase associated with copper; and glutathione transferase) have decreased expression in diabetes; however, enzymes responsible for ROS production in the mitochondrial matrix, such as superoxide dismutase associated with manganese, showed no significant changes in expression. These data suggest that because of the decreased expression of proteins in the ROS pathway in cytosol, it is more susceptible to oxidative damage to mitochondria 17,18. Decreased expression of PRX6 may occur during oxidative stress due to different environmental factors. It has been shown that ROS is an actuator for the latent form of TGF-β and that TGF-β1 inhibits the transcription of PRX6 19. At first glance, our results may be inconsistent with those of other articles published but Li et al through using the techniques of proteomics showed that PRX6 is one of the proteins that has different expression levels in different tumors 20. Ha et al reported that human PRX1 plays a role in possible inhibition of TGF-β 21. Given that PRX1 and PRX6 are members of the same family (peroxide), we can assume that PRX6 has similar effects to PRX2; moreover, because TGF-β probably declined in the treated group, PRX6 declined as well. This inconsistency in the expression of PRX6 may be due to different expressions in different tissues.
Cofilin-1 showed significantly decreased expression in the treated group as compared with that in the normal and diabetic groups. Cofilin binds to F-actin in the cytoskeleton and depolymerizes F-actin in a pH-dependent manner. It plays an important role in the regulation of cell morphology and organization of the cytoskeleton. Regulation of actin dynamics in podocyte activities to maintain glomerular filtration barrier is crucial. In vivo and in vitro studies have shown that cofilin 1 is an essential regulator of actin filament recycling that plays a role in podocyte activity 22. In vitro studies on cultured human and rat podocytes showed that stimulation with TGF-β increased cofilin 1 phosphorylation and decreased its activity 23,24. Our results show that TGF-β inhibition due to fibromodulin decreased cofilin expression.
Tubulin α protein showed a significant increase in expression in treated group as compared with that in the normal and diabetic groups. Tubulin α is one of the main components of microtubules that plays a role in material reabsorption in the kidney tubules via vesicular transport from the surface of the lumen to the tubules baseline 25. Recently, it has been shown that glycosylation of tubulin inhibits its polymerization. The results of the present study show that reabsorption of certain substances in the proximal tubules and their transport by microtubules is disrupted because of tubulin polymerization due to abnormal glycosylation in diabetes 26.
TGF-β1 protein showed significantly decreased expression in the treated group as compared with diabetic groups. TGF-1 is a potent mediator of fibrotic processes through stimulation of the synthesis of extracellular matrix components. TGF-1 also induces albuminuria 27. Elevated renal TGF-β1 mRNA and protein levels have been found in various animal models and the human form of diabetic nephropathy 28. FMOD along with other small leucine-rich proteoglycans such as decorin can interact with a number of different cell surface receptors, cytokines, ECM components, and growth factors, such as TGF-β ligands to modulate their activity 4. Down-regulation of TGF-1 by FMOD indicates that this protein exerts its effect on proteins mentioned above through TGF-1 interaction with some of this proteins and can be a therapeutic target in DN.
Conclusion :
In conclusion, Ad-FMOD has a potential therapeutic role in human DN and Ad-FMOD treatment markedly attenuated the albuminuria in diabetic rats. However, the molecular mechanisms involved in this process require further study.
Acknowledgement :
We thank the Research Deputy of Zanjan University of Medical Sciences (ZUMS) for financial support of this project (MSc thesis, grant number: A-10-349-10).
Conflict of Interest :
The authors declare no conflict of interests.
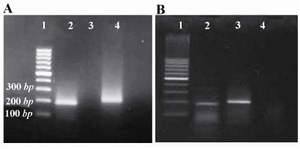
Figure 1. Fibromodolin mRNA detection in HEK293 cells A) and rat kidneys; B) by RT-PCR. A) (Lane 1) 100 bp DNA ladder, (Lane 2) Fibromodulin band (196 bp), (Lane 3) No-template control, (Lane 4) actin band (207 bp).
B) (Lane 1) 100 bp DNA ladder, (Lane 2) Fibromodulin band (196 bp), (Lane 3) actin band (207 bp), (Lane 4) No-template control
|
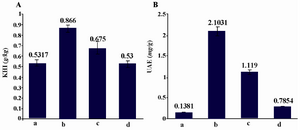
Figure 2. KHI and UAE measurement of infected rats by RAd- FMOD. A) KHI measures in all diabetic control groups, including RAd-lacZ group were higher than those in healthy control group. The KHI of the RAd-FMOD group was slightly lower than that of the diabetic control group. However, there were no significant differences in KHI measures among all diabetic rat groups. B) Measures of UAE in the RAd-FMOD group showed significantly smaller UAE values than the Ad-lacZ group (p0.001).
a) Healthy rats, b) Diabetics rats+saline, c) Diabetic rats+RAd-lacZ, d) Diabetic rats+RAd-FMOD
|
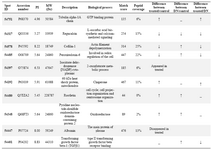
Table 1. MALDI-TOF-MS identification of proteins isolated from kidney treated with RAd-FMOD and diabetic nephropathy kidney
↓; down-regulated, ↑; up-regulated
|
|