Production and Characterization of a Murine Monoclonal Antibody Against Human Ferritin
-
Bayat, Ali Ahmad
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Yeganeh, Omid
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Ghods, Roya
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Zarnani, Amir-Hassan
-
Nanobiotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Bahjati Ardekani, Reza
-
Reproductive Biotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Mahmoudi, Ahmad Reza
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
Mahmoudian, Jafar
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
-
 Jeddi-Tehrani, Mahmood
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran, Tel: +98 21 22432020; E-mail: mahjed@yahoo.com
Jeddi-Tehrani, Mahmood
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran, Tel: +98 21 22432020; E-mail: mahjed@yahoo.com
-
Monoclonal Antibody Research Center, Avicenna Research Institute, ACECR, Tehran, Iran
Abstract: Background: Ferritin is an iron storage protein, which plays a key role in iron metabolism. Measurement of ferritin level in serum is one of the most useful indicators of iron status and also a sensitive measurement of iron deficiency. Monoclonal antibodies may be useful as a tool in various aspects of ferritin investigations. In this paper, the production of a murine monoclonal antibody (mAb) against human ferritin was reported.
Methods: Balb/c mice were immunized with purified human ferritin and splenocytes of hyper immunized mice were fused with Sp2/0 myeloma cells. After four times of cloning by limiting dilution, a positive hybridoma (clone: 2F9-C9) was selected by ELISA using human ferritin. Anti-ferritin mAb was purified from culture supernatants by affinity chromatography.
Results: Determination of the antibody affinity for ferritin by ELISA revealed a relatively high affinity (2.34×109 M-1) and the isotype was determined to be IgG2a. The anti-ferritin mAb 2F9-C9 reacted with 79.4% of Hela cells in flow cytometry. The antibody detected a band of 20 kDa in K562 cells, murine and human liver lysates, purified ferritin in Western blot and also ferritin in human serum.
Conclusion: This mAb can specifically recognize ferritin and may serve as a component of ferritin diagnostic kit if other requirements of the kit are met.
Introduction :
Ferritin is a ubiquitous and highly conserved iron storage protein which plays major roles in iron homeostasis such as protective function against toxic effects of iron overload in cells, cytoprotective antioxidant in pancreatic B-cells and endothelial cells, regulation of the expression of globin genes in erythroid cells, suppression of mRNA translation and proliferation of myeloid progenitor cells 1. This protein (outside diameter 12-13 nm, inside diameter 7-8 nm) is produced by nearly all living organisms and is found in eukaryotes and prokaryotes 2. A 450 kDa ferritin complex consists of 24 subunits of two types, H (heavy; 21 kDa) and L (light; 19 kDa) subunits, and can store up to 4500 iron (Fe3+) ions 3,4. The H-subunit has a relatively acidic electrophoretic dynamism and acts as a ferroxidase that oxidizes iron to (Fe3+) ions, whereas the L-subunit interferes with iron nucleation, mineralization and long term iron storage 2. Ferritin that is not associated with iron is called apoferritin and subunit combination varies in different tissues 5.
High serum ferritin levels have been reported in patients with elevated iron stores, and low levels are associated with iron deficiency 6. Recent studies have reported that perturbations in ferritin levels are associated with the progression of tumor cells in breast cancer 7-9 liver, lung, and prostate cancers 10 by which ferritin perturbations directly instigate tumorigenesis and cause malignant phenotype 7,8. Since the serum of patients with a variety of tumors contains high levels of ferritin 11-13, it seems that the serum ferritin is a good marker for cancer. Therefore, detection of ferritin by mAb as a rapid and sensitive method is necessary 14. Besides, anti-ferritin mAb can be used for designing diagnostic kits for measurement of ferritin in various biological fluids. Production of such kits seems to be more appropriate for patients than other methods for evaluation of iron stores such as liver biopsy, or bone marrow biopsy 15,16. This study aimed to produce and characterize a high affinity murine anti human liver ferritin antibody.
Materials and Methods :
Immunization procedure: Two Balb/c mice (6-8 weeks old) were immunized with purified human ferritin 17. Briefly, each mouse was immunized 5 times with 50 µg of ferritin every 2 weeks. The first immunization was performed using complete Freund's adjuvant. Incomplete Freund's adjuvant was used for subsequent immunizations. One week after the last immunization, blood was collected by a vertical incision of the tail vein followed by determination of antibody titers by ELISA. Finally, three days before the cell fusion, 20 µg of ferritin (without any adjuvant) were injected intravenously 18.
ELISA: Mouse serum titrations and screening of hybridoma supernatants were performed by Enzyme-linked immunosorbent assay (ELISA). The wells of ELISA plate (Nunc, Roskilde, Denmark) were coated with 50 µl of ferritin (10 µg/ml) dissolved in Phosphate Buffered Saline (PBS) and then incubated at 37C for 1 hr followed by overnight incubation at 4C. Then the plates were washed 3 times with PBS containing 0.05% Tween 20 (PBS-T) for 3 min. followed by blocking with 2.5% bovine serum albumin (BSA) at 37C for 1 hr. Wells were then washed 3 times and mouse sera (in two-fold serial dilutions starting from 1:500) were added and incubated for 1 hr and wells were again washed with PBS-T. Rabbit anti mouse Ig conjugated to horseradish peroxides (1:1000) (Avicenna Research Institute, Tehran, Iran) were added to the wells and incubated for 1 hr at 37C. After washing, 50 µl of tetramethylbenzidine (TMB) (Sigma-Aldrich, Missouri, USA) substrate was added to each well and the plates were incubated at room temperature in the dark. After 15 min, the reaction stopped by adding 15 µl of stop solution (20% H2SO4) to each well. The Optical Density (OD) of the reactions was measured at 450 nm by an ELISA reader (BioTek, Winooski, VT, USA). The mouse with higher titer of antibody was selected for fusion. To screen the antibody production of hybridoma cells, the same method was done on the cell supernatants.
Hybridoma cell production: Mouse myeloma Sp2/0 cells, used as fusion partners, were cultured and propagated in RPMI-1640 culture medium (Gibco, Gran Island, NY, USA) and 10% Fetal Bovine Serum (FBS) (GIBCO Invitrogen, USA). Spleen cells from the immunized mouse were mixed with the Sp2/0 cells at a ratio of 1:5 (1 Sp2/0 and 5 spleen cells). The mixture was washed twice with pre-warmed RPMI-1640 (37C). Then, pre-warmed 50% polyethylene glycol (PEG) 1500 (Sigma-Aldrich, Germany) was used for fusion. Selective HAT medium (Sigma-Aldrich, Germany) was then used for selection of hybridoma cells. The reactivity of culture supernatants was then tested by ELISA 18. Finally, positive hybridomas were cloned by limiting dilution process 19.
Antibody purification: Anti-ferritin mAb 2F9-C9 was purified from culture supernatants by affinity chromatography using a Hi-Trap protein G column (GE Healthcare, Uppsala, Sweden). Briefly, culture supernatants were filtered through 0.45 µm filters and pH was adjusted to 7.5. The elution was performed using Glycine-HCl (0.1 M, pH=2.7). The eluted antibody was dialyzed against PBS at pH=7.5 and the reactivity of the purified antibody was determined by ELISA method as mentioned above.
Isotype determination: Goat anti mouse IgG1, IgG2a, IgG2b, IgG3, IgA and IgM (Sigma-Aldrich, Missouri, USA) at 1/1000 dilution were coated in the wells of ELISA plate (Nunc). Supernatant of the growing hybridoma 2F9-C9 was added to each well. The isotype of 2F9-C9 mAb was then determined according to the ELISA method described above.
Detection of ferritin in human serum with anti-ferritin 2F9-C9 mAb by direct sandwich ELISA: To determine the reactivity of 2F9-C9 with free ferritin and ferritin in human sera, 2F9-C9 mAb (50 μl/well, 5 μg/ml) was coated in a 96 well plate and then, different dilutions of ferritin from 0 to 250 ng/ml as well as human sera (1:5 dilution) were added (50 μl/well). HRP-conjugated anti-ferritin polyclonal antibody (Avicenna Research Institute, Tehran, Iran) was then added. After washing, TMB was added as substrate and the test continued as described in ELISA section. For comparison, 1:5 diluted human sera were also tested by a commercial ferritin kit (RADIM, Florence, Calenzano, Italy).
Flow cytometry: Hela (Human cervical carcinoma) cells were harvested by 0.5% trypsin and 0.1% EDTA (Gibco) and permeabilized by permeabilizing solution (Becton Dickinson, USA). Cells were then blocked with 5% sheep serum for 10 min, and then cells were incubated with 100 µl of 2F9-C9 mAb (10 µg/ml) for 1.5 hr at 4oC. Anti-HIV Env (Avicenna Research Institute, Tehran, Iran) with IgG1/К isotype was used as an isotype control 20. After 3 times of washing with cold PBS, FITC-conjugated sheep anti-mouse Ig (Avicenna Research Institute, Tehran, Iran) (1:50) was added to cells and incubated for 45 min in the dark at 4oC. Cells were washed and sorted by flow cytometry. The data was analyzed using Flowmax software (Partec, Nuremberg, Germany).
Western blotting: Lysates of the cell lines Raji (human burkitt's lymphoma(, PC3 (human prostate cancer), K-562 (human myelogenous leukemia), human and mouse liver lysates (lysis buffer: 150 mM NaCl, 1 mM EDTA, 50 mM Tris HCl pH=7.4, 1% Triton X-100, 1% Sodium deoxycholate, 0.1% SDS), supplemented PI (phosphatase inhibitor) (Roche, Basel, Switzerland) and 1% PIC (protease inhibitor cocktail) (Sigma-Aldrich, Missouri, USA) were prepared. The protein concentrations of the lysates were measured by BCA protein assay kit (Thermo Scientific, Rockford, IL. USA). Twenty g of cell lysates and 200 ng purified ferritin were run on a 15% SDS–PAGE gel.
After electrophoresis, resolved proteins were transferred onto PVDF membranes (Millipore Corporation, Billerica, Mass chusetts, USA). The membranes were blocked with 5% non-fat milk in PBS-T overnight at 4C. After gentle washing with PBS-T, 2F9-C9 mAb (5 µg/ml) was added to the membrane and incubated for 1.5 hr at room temperature. The membrane was washed extensively with PBS-T and incubated with HRP-conjugated Rabbit anti-mouse Ig (Avicenna Research Institute, Tehran, Iran) (1:2500) for 1 hr at room temperature followed by washing and developing with ECL Chemiluminescence detection system (GE Healthcare).
For validation of protein band specificity detected in Western blot, reactivity of anti-ferritin mAb was blocked with a saturating concentration of ferritin (30:1 ferritin to antibody molar ratio). In this regard, ferritin was added to 2F9-C9 mAb for 1 hr at 37C and then the mixture was added to PVDF membrane. Unblocked anti-ferritin mAb was added to another PVDF as a positive control. The incubations, washings and development of bands were performed as the above 21.
Determination of affinity constant (Kaff): The affinity constant (Kaff) of 2F9-C9 mAb was determined by ELISA 21,22. Briefly, different concentrations of ferritin (5000, 2500, 1250, 625, 312.5, 156, 78 and 39 ng/ml) were coated in 96 well ELISA plates. Serial dilutions of 2F9-C9 mAb (5000, 1250, 312.5, 78, and 19.5 ng/ml) were added to each coated well. Sigmoid curves were plotted using the OD values obtained for different concentrations of mAb. The affinity constant was measured as described elsewhere 23.
Results :
Production of anti-ferritin mAb: After immunization of mice with ferritin, the titers of anti-ferritin antibodies in the mice sera were measured by ELISA. Results showed that mouse 1 had higher titer of anti-ferritin Ab (Figure 1). After fusion between splenocytes of mouse 1 and Sp2/0 cells, supernatants of growing hybridoma cells were screened based on reactivity with ferritin by ELISA. In this screening assay, among 20 positive clones, a clone was characterized as 2F9-C9 with high reactivity with ferritin (OD: 1.438). The isotype of 2F9-C9 mAb was determined to be IgG2a (Figure 2) and its affinity was calculated to be 2.34×109 M-1 (Figure 3) and (Table 1).
Detection of ferritin in human serum: Known concentrations of ferritin were used to plot ELISA standard curve for 2F9-C9 mAb (Figure 4A). ELISA standard curve for the commercial ferritin measurement kit was also plotted according to the manufacturer's instructions (Figure 4B). As shown in table 2, the concentrations of ferritin in 5 human sera which were measured with our ELISA test using 2F9-C9 mAb and the commercial kit were found to be very similar.
Flow cytometric properties of 2F9-C9 mAb: Since Hela cell line expresses ferritin intracellularly, 2F9-C9 mAb was applied for intracellular staining of ferritin in this cell line. Figure 5A shows that 2F9-C9 mAb recognized intracellular ferritin molecules in 79.4% of Hela cells, while the ferritin negative HepG2 cell line showed no ferritin expression by flow cytometry using 2F9-C9 (Figure 5B).
Western blot analysis: Western blot analysis demonstrated that 2F9-C9 mAb recognized ferritin as an approximately 20 kDa protein band in human liver, mouse liver and K-562 cells (Figure 6A). Additionally, blocking of the antibody with human ferritin resulted in abrogation of its reacting, hence no band was detected in western blot (Figure 6B).
Discussion :
Producing a high affinity monoclonal antibody against human ferritin provides a convenient means for monitoring and tracking ferritin in human body fluids. In this study, a murine anti-ferritin mAb with a high affinity constant was produced and characterized. 2F9-C9 mAb recognizes a specific protein band around 20 kDa in Western blot and blocking of the antibody with purified human ferritin led to disappearance of the protein band, confirming the specificity of 2F9-C9 mAb for ferritin. On the other hand, 2F9-C9 mAb recognized intracellular ferritin molecules in 79.4% of Hela cells whereas HepG2 cells were negative. Besides, mAb identified ferritin in some human serum samples in a similar manner to a commercial ferritin measurement kit that also confirms mAb specificity for ferritin. In addition, human serum ferritin contains nearly only L subunits 24 and considering the fact that H and L chains of ferritin have about 60% amino acid sequence homology 3,25,26, 2F9-C9 mAb probably recognizes the L chain or a common epitope on the L and H chains. Human and mouse ferritin have 93% homology in theprotein sequences (NCBI, blastp). Considering the high degree of homology between human and mouse ferritin molecules, it is most probable that the antibody recognizes a shared epitope on both molecules.
To compare with other studies, Lee et al reported that only one out of more than 1000 hybrid clones had a high affinity (1×1010 M-1) for human ferritin and by using this antibody they showed that under identical conditions, purified ferritin from human spleen and human heart showed 50 and 30% cross-reactivity, respectively 14. Mel'nikova et al also obtained three mAbs with affinity constants ranging from 6×108 to 3×109 M-1. They used these mAbs to define their binding sites on a ferritin molecule. They concluded that only four IgG molecules could simultaneously bind a ferritin molecule 27. In addition, Nozawa et al developed an anti human placental ferritin mAb which was used to measure serum ferritin in three groups: normal women, normal pregnant women and women with gynecological cancers. They concluded that ferritin was a good marker for cancer research 28. Importantly, ferritin measurement has been applied to study some types of cancers. In this regard, Uesaka produced an anti human liver ferritin that was used to show that serum ferritin levels significantly increased in pancreatic cancer and hepatoma compared to that in normal sera 12.
Conclusion :
In conclusion, anti human ferritin mAb 2F9-C9 was shown to react with a 20 kDa ferritin subunit. The IgG2a antibody was also shown to be capable of detecting serum ferritin by ELISA and intracellular ferritin by flow cytometry.
Acknowledgement :
This study was supported by a grant from Avicenna Research Institute.
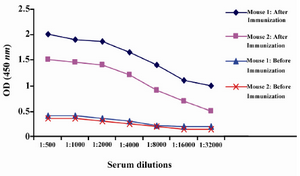
Figure 1. Serum titration of two immunized Balb/c mice by ELISA. Mice were immunized five times by intraperitoneal injection of 50 µg human ferritin
|
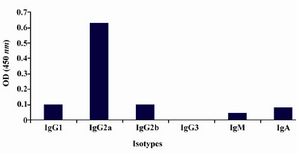
Figure 2. Isotype determination of 2F9-C9 mAb by ELISA
|
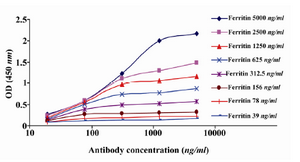
Figure 3. Determination of affinity constant of 2F9-C9 mAb (Kaff) by ELISA. Different concentrations of 2F9-C9 were tested against serial dilutions of human ferritin and Kaff was calculated
|
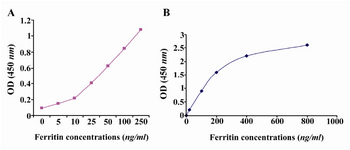
Figure 4. A) In house developed ELISA standard curve for 2F9-C9 mAb; B) Standard curve of commercial kit
|
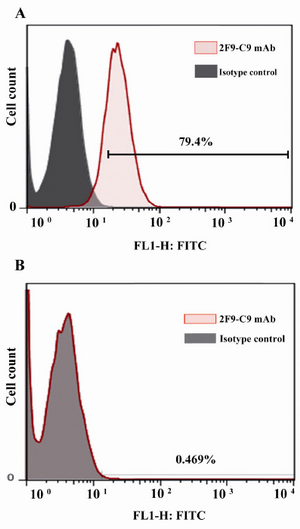
Figure 5. Flow cytometric analysis of intracellular ferritin expression in Hela; A) and HepG2; B) cells using 2F9-C9 mab
|
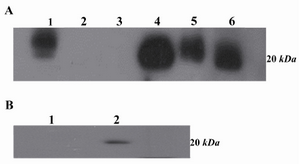
Figure 6. A) Western blot analysis of 2F9-C9 mAb reactivity with different cell lysates and purified ferritin. Lanes 1 and 4 represent human and murine liver lysates, respectively; lanes 2 and 3 include Raji and PC3 cell lysates as negative controls, respectively. Lane 5 includes K-562 cell lysate and lane 6 includes purified human ferritin; B) Lane 1 represents inhibition of 2F9-C9 reactivity with ferritin by saturating amounts of exogenous ferritin. Lane 2 represents unblocked 2F9-C9 reactivity with purified human ferritin
|
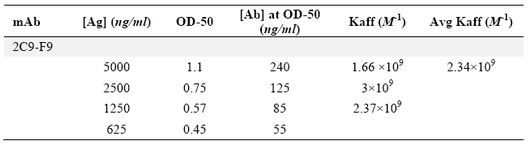
Table 1. Calculation of 2F9-C9 affinity constant
|
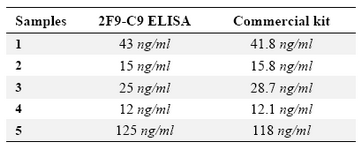
Table 2. Determination of ferritin concentration in 5 human serum samples by in house designed ELISA using 2F9-C9 mAb compared with a commercial ferritin measurement kit
|
|