Combined Treatment of Androgen-Independent Prostate Cancer Cell Line DU145 with Chemotherapeutic Agents and Lithium Chloride: Effect on Growth Arrest and/or Apoptosis
-
 Hossein, Ghamartaj
Department Animal Physiology, Developmental Biology laboratory, School of Biology, University College of Science, University of Tehran, Iran, Tel: +98 21 61112622; Email: ghossein@khayam.ut.ac.ir
Hossein, Ghamartaj
Department Animal Physiology, Developmental Biology laboratory, School of Biology, University College of Science, University of Tehran, Iran, Tel: +98 21 61112622; Email: ghossein@khayam.ut.ac.ir
-
School of Biology, University College of Science, University of Tehran, Tehran, Iran
-
Azimian Zavareh, Vajihe
-
School of Biology, University College of Science, University of Tehran, Tehran, Iran
-
Department of Regenerative Medicine, Royan Institute for Stem Cell Biology and Technology, Tehran, Iran
Abstract: Hormone-independent prostate cancer cell lines are resistant to antineoplastic drugs, this study sought to determine the usefulness of lithium chloride as an inhibitor of glycogen synthase kinase-3β to increase the cytotoxic effect of doxorubicin, etoposide or vinblastine antineoplastic drugs on DU145 cells. Combination effect was assessed by using low and IC50 doses of drugs + lithium chloride. Subsequently, cell cycle analysis and p53 levels and its subcellular localization as a key regulator of cell cycle were assessed. Lithium chloride showed cytotoxic effect in a dose and time dependent manner (p<0.001). Both drugs doxorubicin and etoposide in combination with lithium chloride showed higher percent of cells in SubG1 compared to control (p<0.001). Combination of IC50 dose of doxorubicin and lithium chloride led to S phase arrest (p<0.001, compared to control, lithium chloride or doxorubicin alone). Moreover, G2/M arrest was significantly increased when low dose of doxorubicin and vinblastine were combined with lithium chloride (p<0.001, compared to control and lithium chloride alone). DU145 cells were highly sensitive to vinblastine and no significant changes were observed when combined with lithium chloride. The IC50 doses of all three drugs combined with lithium chloride demonstrated decreased cell percent in G1 phase compared to control or lithium chloride alone (p<0.001). Moreover, in the presence of lithium chloride there were increased levels of p53 in cytoplasm and nucleus (p<0.05). Our results suggest that combination of lithium chloride with chemotherapeutic agents may increases their cytotoxic effect on hormone non-responsive human prostate cancer cells.
Introduction :
Hormone-independent prostate cancer is resistant to a broad range of antineoplastic agents which may be caused by the resistance to induction of apoptosis playing a major role in the occurrence of prostate cancer (1). Therefore, new strategies for cancer therapy focus on enhancing the effectiveness and safety of antineoplastic drugs such as combination therapy with different type of drugs which may be target key kinase players in prostate cancer progression. Glycogen synthase kinase- 3 (GSK-3) was shown to enhance Androgen Receptor (AR) function (2,3). It was demonstrated that exposure of prostate cancer cells to lithium chloride (LiCl), a known GSK-3 inhibitor, abrogated the stimulatory effect of GSK3 on AR-mediated transactivation (2,3). Although, it should be noted that these findings remain controversial given contradictory reports that GSK3 is a negative regulator of AR-mediated transcription (4-6). Subsequently, it was confirmed that LiCl can also increase AR-mediated transcription in the human CWR22rv-1 cell line that also endogenously expresses AR (7). Recent study reported that high levels of activated GSK3β known as pGSK-3βY216 was associated with aggressive PCa (2), and is a critical determinant in the progression of PCa (8). Moreover, inhibition of GSK-3β activity sensitized PCa cells to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-mediated apoptosis (9), confirming the role of GSK-3β as a survival factor (10).
DU145 cell line expresses detectable levels of the AR mRNA and protein but is considered as androgen non-responsive cells (11). Moreover, DU145 cell line are Bax negative (12) harbors a temperature sensitive (TS) mutant of p53 gene (13) which render these cells Fas-resistant (12). Suppression of GSK-3β activity has been reported to reduce proliferation of DU145 human prostate androgen-independent cell line and other hormone responsive PCa cell lines (3,14). Correspondingly, a recent study demonstrated that LiCl significantly suppressed tumor development and growth of PC-3 (androgen non-responsive) and C4-2 (androgen responsive) cells in nude mouse xenograft models (15) which may suggest GSK-3 inhibition as a therapeutic strategy for prostate cancer intervention.
Cytotoxic chemotherapy is being used to control and treat PCa but remains relatively non-selective and highly toxic to normal tissues. In an effort to develop effective strategies that increase the therapeutic potential of cytotoxic anticancer drugs with less systemic toxicity in recent years, more efforts are being directed toward combination chemotherapy (16). Ideally, the combined drugs should have proven single cytotoxic activity, minimal overlapping toxicities and different modes of action. It is anticipated that PCa cells arrested in S-phase will be more sensitive to other cytotoxic drugs (17,18), since LiCl induced S phase arrest in PCa cell lines (14), this promoted us to use it in combination with antineoplastic drugs. The anthracycline antibiotic doxorubicin, (Dox) is a cell cycle non specific drug which may cause cell cycle arrest in different cell cycle phase (19). While, etoposide (Eto) is a semi synthetic derivative of the podophyllotoxins, which inhibits DNA synthesis by inhibiting DNA topoisomerase II which is cell cycle dependent and phase specific, affecting mainly the S and G2 phases (19). Vinblastin (Vin) is a vinca alkaloid which binds tubulin, thereby inhibiting the assembly of microtubules and is M phase cell cycle specific agent (19).
The aims of this study were three folds: 1) to assess the sensitivity of Du145 cells to LiCl, 2) as DU145 have been reported to be resistent to Dox and Eto (1) we sought to determine whether the cytotoxic effects of Dox and Eto on these cells would be modulated in combination with LiCl and also by using Vin to determine whether cell cycle specificity of drugs may be a determinant factor for their selection in combination therapy with LiCl, and 3) finally as DU145 harbors a TS p53 protein, it was interesting to elucidate whether LiCl may have an effect on the
p53 protein stability.
Materials and Methods :
Cell lines and reagents: Human prostate carcinoma DU145 cells were obtained from National Cell bank of Iran (NCBI) affiliated to Pasteur Institute of Iran and grown in RPMI 1640 supplemented with 10% fetal bovine serum and antibiotics at 37°C in a 5% CO2 atmosphere under 90–95% humidity. LiCl and sodium chloride (NaCl) (Merck, Germany), 3-(4, 5-dimethylthiazol-2-yl) 2, 5-diphenyl-tetrazolium bromide (MTT) and propidium iodide (Sigma-Aldriche, USA). RNase A was purchased from iNtRON Biotechnology (Korea). Antineoplastic drugs were obtained from Iranian Red Cross Pharmacy.
Cytotoxicity and anticancer assay: The IC50 of lithium and drugs on DU145 cells was measured by MTT-based cell proliferation assay, which was calculated by using linear regression test of Graphpad Instate-3 software (LaJolla, CA). To this order, DU145 (15000 cells/well) cells were seeded in 96-well plates and next day were treated with either RPMI 1640 alone as a control, or with different doses of LiCl (2.5, 10 and 25 mM) DU145 cells. In parallel, cells were treated with Dox (0, 5, 10, 25, 100, 250, 500 nM), Eto (0, 2.5, 5, 10, 50 and 100 µM) and Vin (0, 0.25, 0.5, 1.0, 2.5, 5.0 and 10 nM) alone, or in combination with LiCl for 24, 48 and 72 hr. At the end of exposure, 10 μl of MTT (Sigma, USA) stock solution (5 mg/ml) was added to 100 μl of medium in each well and plates were incubated for 4 hr at 37°C, subsequently MTT-formazan product was dissolved by adding 100 μl of acidic isopropanol to each well. The plates were incubated about 10 min at room temperature before determination of absorbance (A) values at 570 nm with Elisa reader. Cell survival was expressed as percentage (A value of treated to control cells ratio x 100). Experiments were performed in triplicate and repeated at least three times. To rule out the effect of the anion (Cl-) through an osmotic disturbance, we compared LiCl with NaCl at same molar concentrations.
Flow cytometry assay: Cell cycle distribution and measurement were performed by flow cytometry. To this order, cells (75×105 cell/ml) were serum-starved for 24 hr and were returned to serum-containing media with LiCl and drug either alone or in combination for 48 hr. After treatment, pool of floating cells in the medium and harvested attached cells were washed with cold Phosphate-Buffered Saline (PBS) and fixed in 70% ethanol (-20°C) at 4°C. After 2 hr, fixed cells were pelleted and stained with propidium iodide (20 μg/ml) in the presence of RNase A (100 g/ml) for 30 min at 37°C, and about 104 cells were analyzed using a fluorescence activated cell sorter. Data were analyzed by using WinMDI 2.9 software.
Immunocytochemistry: In order to detect p53 protein we used a mouse monoclonal anti-p53 antibody (p53, (DO-1), Santa Cruz Biotechnology, Inc. USA) which recognize all forms of p53 wild type and mutated forms. Cells were plated in 96 well plates and then treated with IC50 dose of LiCl and drugs alone or in combination for 48 hr. Briefly, cells were washed with PBS prior to fixing with 1% glutaraldehyde for 15 min at room temperature. Subsequently, non specific binding sites were blocked with 3% normal goat serum in 1% BSA for 30 min; then cells were incubated with anti-p53 monoclonal antibody (1: 200) diluted in 1% BSA + 0.1% Triton ×100, overnight at 4C. p53 immunoreactivity was visualized by exposing the cells to HRP LSAB reagents (Dako) and revealed with diaminobenzidine tetra hydrochloride (DAB). The cells were counterstained with hematoxylin, rinsed and mounted with Vectamount (Vector laboratories. CA, USA). Negative controls were carried out by omitting the primary antibody.
Western blot analysis: DU145 were seeded in 6 wells plates at 500×103 cells/well treated without or with antineoplastic drugs or LiCl alone or in combination for 48 hr, then homogenized by using RIPA buffer (50 mM Tris, pH=8.4, 150 mM Nacl, 1% Triton X-100, 0.5% Sodium deoxycholate, 1% SDS, 5 µg/ml protease inhibitor cocktail I, 5 µg/ml protease inhibitor cocktail II, 1 mm sodium fluoride, and 1 mm Sodium ortho vanadate and centrifuged (13000 rpm) at 4oC. Supernatants were removed and the amount of protein per sample was determined by Bradford test. Cell extracts (20 µg) were run on SDS-PAGE and transferred to PVDF membranes (Bio-Rad laboratories, Inc., CA, and USA) by electrophoresis. Membranes were blocked with 3% BSA (Merck, Germany) with agitation at room temperature for 1 hr, followed by incubation with mouse monoclonal anti-p53 antibody (Santa Cruz Biotechnology) diluted 1:100 in 1% BSA overnight at 4oC, washed three times with Tris-buffered saline and incubated for 1 hr at room temperature with anti-mouse IgG peroxidase-linked antibody (Sigma-Aldrich, USA). Next, membrane was washed extensively with Tris buffer containing 0.5% Tween 20 and detection of immunoreactive protein was performed by using enhanced chemiluminescent (ECL) detection reagents (Millipore) followed by autoradiography.
In order to quantify the amount of immunoreactive protein, blot was stripped with stripping buffer (150 mg/ml Glycin, 10 mg/ml SDS, 10 µl/ml Tween 20, pH=2.2,) for 10 min, blocked and incubated with rabbit polyclonal anti β-actin antibody (Sigma-Aldriche, USA) diluted 1:1000 for 1 hr at room temperature. Blots were washed extensively in Tris-buffered saline containing 0.5% Tween-20, and incubated with anti-rabbit IgG peroxidase-linked antibody (Bio-rad, laboratories, Inc., CA, USA) diluted 1:30,000 in 1% BSA and detected with ECL reagents. Band density was quantitated by densitometric analysis using Scion program. The full-scale ordinate value was set at 1.0 absorbance unit.
Statistical analysis: Nominal variables were analyzed by the use of the Kolmogorov Smirnov test. Skewed and normal distributed metric variables were analyzed by Mann-Whitney U and one-way Anova tests, respectively. Analysis of combined growth-inihibitory effect was performed by using two way Anova. p≤0.05 was considered to be statistically significant. All statistical procedures were run on SPSS version 16 (SPSS Inc., Chicago, IL).
Results :
LiCl effect on DU145 cells growth: DU145 cells viability in the presence or absence of LiCl (2.5 to 25 mM) was assessed as percent of viable cells compared to control (in the absence of LiCl). Cells showed 32% (p<0.05), and 53% (p<0.001) reduced cell viability with 10 and 25 mM LiCl after 48 hr (Figure 1A). A significant decreased cell viability of 13% (p<0.05) was observed with low as well as with high dose of LiCl (55%, p<0.001) after 72 hr. These results showed that sensitivity of DU145 cells to LiCl was time dependent (Figure 1A). No significant effect was observed with NaCl (2.5 to 25 mM) as control (Figure 1B). In the next step in order to define the usefulness of antineoplastic drugs and LiCl in combination therapy, cytotoxic assay was performed first with drugs alone, and then with LiCl in combination. LiCl IC50 dose for 48 hr treatment was 20 mM.
Effect of drugs alone or in combination with LiCl on DU145 Cells growth: Dose response curves for each drug were shown in figure 2. Dox had no cytotoxic effect on DU145 cells after 24 hr, and showed a significant cytotoxic effect after 48 and 72 hr with all doses of Dox (p<.001), (Figure 2A). Eto had no effect on DU145 cells after 24 hr, however cell viability reduction was observed with 2.5 to 100 M after 48 hr (p<0.05 and p<0.001, respectively) and 72 hr (p<0.001) (Figure 2B). However, Vin showed a strong and significant cytotoxic effect on DU145 cells from low dose of 2.5 nM to 10 nM (Figure 2C).
Next we sought to determine whether antineoplastic drugs in combination with LiCl may increase the sensitivity of these cells to these drugs. Based on obtained results with different timing, the selected time for these experiments was 48 hr as a best time for assessment of combination therapy on cells viability. Two different doses of drugs; 1) IC50 doses of 230 nM, 18 M and 2.5 nM for Dox, Eto and Vin, respectively, or 2) low doses of 25 nM, 2.5 M and 1.5 nM for Dox, Eto and Vin, respectively. Each concentration of drug was used alone or in combination with IC50 dose (20 mM) of LiCl.
Results of cell viability is shown for combination effect of Dox and LiCl in figure 3A. Dox at 25 nM dose caused 38% decreased cell viability (p<0.05 compared to control). However, when combined with 20 mM LiCl this showed 47.5% cell survival inhibition (p<0.01 compared to control); comparable results was obtained with 20 mM LiCl alone (45.5% decreased cell viability p<0.001 compared to control). With Dox IC50 dose (230 nM) 48.6 % cell viability reduction was observed (p<0.001) which was most effective in combination with 20 mM (68.8% inhibition, p<0.001 compared to control; p<0.05 compared to LiCl alone). Thus, use of either low or effective doses of Dox and LiCl in combination were more effective and induced a highly significant cytotoxic effect compared to each substance alone (Figure 3A). Most importantly, cytotoxic effect of low dose of Dox combined with LiCl, was similar to IC50 dose of Dox alone (Figure 3A).
For combination effect of Eto plus LiCl, a low dose of Eto (2.5 µM) and IC50 dose (18 µM) alone or in combination with LiCl was used. As shown in figure 3B, 2.5 µM Eto showed significant effect on cells viability (24% reduction compared to control p<0.05), and comparable higher decreased in cell viability was obtained when combined with 20 mM LiCl, (42.5%, p<0.01 and p<0.05 compared to control and Eto 2.5 M alone, respectively). Treatment of cells with 18 µM Eto alone showed 47% cell growth inhibition (p<0.001) and its combination with LiCl revealed a highly significant cell growth inhibitory effect of 63.5% compared to control (p<0.001), Eto 18 M (p<0.05) or LiCl alone (p<0.05). Altogether, these results showed a higher significant cytotoxic effect of low dose of Eto combined with LiCl.
Vin had a high cytotoxic effect on DU145 cells with low dose of this drug and the difference between low and effective concentration of this drug was minimum compared to two other drugs. As revealed in figure 3C, cell growth inhibition were 44% (p<0.05) and 54%, (p<0.001) for 1.5 or 2.5 nM concentrations, respectively. The combined effect of 1.5 nM Vin plus 20 mM LiCl led to 59% reduced cell viability which was highly significant compared to control (p<0.01), and drugs alone (p<0.01). Comparable cell growth inhibition (62%, p<0.001 compared to control) was obtained with IC50 doses of Vin + LiCl in combination (Figure 3C). However, cytotoxic effect of low dose of Vin with LiCl was more effective compared to low dose of Vin alone (59% versus 34%, p<0.01, respectively).
Effect of LiCl-antineoplastic drugs combination on cell cycle progression in DU145 cells: To determine whether the drug + LiCl-induced inhibition of cell proliferation was due to altered cell cycle regulation, DU145 cells were serum-starved for 24 hr and then treated with LiCl and drugs alone or in combination for 48 hr. Cell cycle profiles were monitored by flow cytometric analysis of DNA content in the absence or presence of IC50 and low doses of each drug alone or in combination with IC50 dose of LiCl.
Du145 cells treatment with LiCl showed a decrease of cells population in G1 (p<0.001, compared to control) and an increase in cell percentage in S and G2 phases, although not significant (Figure 4, Table 1). Whereas, low dose of Dox had no significant effect on cells distribution in interphase, however, when combined with LiCl a highly significant (p<0.001) decrease of cells percent in G1 was observed compared to control, and LiCl or Dox alone, followed by cells arrest in G2/M (p<0.001, compared to control; p<0.05 compared to LiCl alone) (Figure 4, Table 1).
Comparable trend was observed with IC50 dose of Dox alone or in combination with LiCl which means decreased percent of cells in G1 and cells arrest in S and G2/M (Figure 4, Table 1). It should be noted that cells arrest in S phase is significantly different from control and LiCl alone (p<0.001) and also compared to IC50 dose of Dox (p<0.01) (Figure 4, Table 1). Although treatment of cells with LiCl or Dox alone increased apoptosis as revealed by increased subG1 cell population, this increase was only significant with effective dose of Dox alone (p<0.05, compared to control) or combined with LiCl (p<0.001, compared to control) (Figure 4, Table 1).
Low or IC50 doses of Eto caused S phase arrest which was only significant with 18 M Eto. Interestingly, low concentration of Eto 2.5 M led to significant increased G2/M arrest (Figure 5, Table 2). Cells treated with two different doses of Eto showed increased apoptosis compared to control (p<0.001). The important point is that increased percent of cells in subG1 was significantly higher with IC50 dose of (Eto + LiCl) compared to control or LiCl alone (p<0.001) (Figure 5, Table 2).
It should be noted that cell growth inhibition with either 2.5 or 5 nM Vin we observed almost 70% decreased cell proliferation (data not shown). Thus, in order to have a better understanding of Vin effect on cell cycle, experiments were designed with 1.5 and 5 nM to have a greater difference between doses. Decreased percent of cells in G1 phase (p<0.001) and G2/M arrest (p<0.001) were found with 5 nM Vin alone compared to control (Figure 6, Table 3). Although G2/M arrest was observed with the two different doses of Vin in combination with LiCl, the percent of cells in this phase was higher with 5 nM Vin + LiCl which was significant compared to control and LiCl alone (Figure 6, Table 3). Vin similar to two other drugs increased apoptosis as revealed by higher percent of cells in subG1 (Figure 6, Table 3). However, it should be stressed that Vin combination with LiCl attenuated the cytotoxic effect as the percent of cells in subG1 were lesser compared to drug alone (Figure 6, Table 3).
Subcellular localization and western blot analysis of p53: It is well known that DU145 harbors temperature sensitive p53 protein due to two different mutations in both alleles of p53 gene (13) which was reported to have anti-Fas activity in human prostate cancer cell lines (20). Important literature reports the important role of p53 in cell cycle checkpoint and apoptosis and tumorogenesis (21), of note LiCl could stabilize p53 in endothelial cells (22) but this possibility has not been investigated in DU145 cell line. Since we found evidence of higher apoptosis rate in cells treated with Dox and Eto in combination with LiCl, we were tempted to analyse first the subcellular localization of p53 and quantify its amount by western blot analysis. DU145 showed distinct punctate cytoplasmic staining with a nucleus component. Moreover, result showed a greater immunostaining in cells treated with LiCl alone compared to control (Figure 7) which showed to be significant when quantified by using a software using C language (Microsoft Visual Studio 2005 and Dot Net framework 2.0) was used to perform the image processing (23). This result was confirmed by protein analysis which also showed an increase of p53 levels (p<0.05) with LiCl compared to control (Figure 8).
Discussion :
Hormone-independent prostate cancer is resistant to a broad range of antineoplastic agents (24). However, it seems that its most acute manifestation is the failure to treat metastasis. A part of this problem is the imperfect effectiveness of adjuvant chemotherapy as the tool to eradicate undetectable micrometastases. In view of toxicity of anticancer drugs, optimal scheduling is potentially useful in improving these treatments. In this regard, using enzymes that could interfere with AR, PI3K/Akt or Wnt signaling activities which were shown to play key role in prostate cancer progression would be an interesting therapeutic strategy (25). One of the most important enzyme is GSK-3 that its involvement has been reported in androgen-stimulated gene expression (2,5). Thus, it is of interest to know whether there is any additive cytotoxic effect of antineoplastic drugs when combined with LiCl on DU145 cells. To answer this question, we examined cytotoxic effect of LiCl or drugs alone or in combination by assessment of cell cycle analysis and also to determine their effect on p53 levels as an important protein involved in cell cycle checkpoints and apoptosis. Here we report for the first time the usefulness of LiCl and antineoplastic drugs combination to increase growth arrest and/or apoptosis of a hormone non-responsive PCa cell line.
GSK-3β is able to regulate many signaling pathways, because it possesses unique structural characteristics that causes it to recognize pre-phosphorylated substrates (26). Moreover, GSK-3 and AR co-distribute in immunohistochemical staining of prostate cancer tissue samples which showed that GSK-3 and AR were diffusely distributed in the cytoplasm or concentrated in the nuclei of prostate cancer cells (5). Meanwhile, cytoplasmic accumulation of GSK-3β protein in prostate cancers was found to correlate with disease progression (27). Furthermore, aberrant GSK-3β activation (Y216 phosphorylation) was shown in highly aggressive prostate cancer cells (2). Consistently, suppressing GSK-3 activity reduced prostate cancer cell proliferation in vitro (3,14,28).
The co-distribution and also co-immunoprecipitation of GSK-3 and AR in prostate cancer tissue suggests that these molecules are capable of interacting in both the cytoplasmic or nuclear compartments of prostate cancer cells, suggesting that these two molecules may interact during the progression of prostate cancer. Indeed, both activated or inhibited form of GSK3 phosphorylation on Y216 or serine 9 detected in DU145 cells, respectively (2,5).
However, controversy may still exist on the role of GSK3β on AR activity, as one study showed active GSK3β is required for inhibition of AR activity (5). While other studies support firm evidences for the role of GSK3β activity as a required enzyme for AR activity (3,14,28). In addition, it has been shown that AR is necessary for G1 to S transition (17) and previous report showed that LiCl caused S phase arrest of DU145 cells by reduction of E2F-mediated gene expression and reduced cdc25C expression, a protein phosphatase required for S-phase completion and mitosis entry (14). In agreement with previous report (14), the present study showed the anti-proliferative effect of LiCl on DU145 cells.
Here, the main findings regarding combined effect of Dox and LiCl were increased cytotoxic effect of low dose or IC50 dose of Dox in combination with LiCl, which was related to cell cycle alteration (Table 1). Low dose of Dox combined with LiCl caused significant decrease of cell population in G1 phase and high G2/M arrest. While, combination of IC50 dose of Dox and LiCl in addition to decreased cell number in G1 phase showed also a significant S phase arrest and apoptosis (Table 1). Dox is a cell cycle non specific drug which may cause arrest in different cell cycle phase (19) and inhibits DNA synthesis by inhibiting DNA topoisomerase II, intercalates with DNA and directly affect transcription and replication (29,30). It has been demonstrated that Dox also activates p53–DNA binding which causes the induction of Cip1/p21 and results in the G1 arrest in cells with wild type p53 protein (30). Here, DU145 with mutant p53 were not arrested at G1 phase, progress through to the S phase and became sensitive to Dox where the expression of α-isoform of topoisomerase II is increased during DNA synthesis (30).
The interest of LiCl use is that it induced S phase arrest of human PCa cell lines (14), correspondingly, here we found S phase arrest with LiCl and it is believed that cells in S phase are more sensitive to chemotherapeutic agents (17,18). Recruitment of cells into S phase, has been used as a strategy to increase drug incorporation into cells. Thus, higher apoptotic effect of LiCl and Dox may result from LiCl-induced cell cycle arrest in S phase, thereby exposing a higher proportion of tumor cell population to the drugs during active DNA replication. As Dox has serious side effects, use of LiCl combined with lower dose of this agent may be an interesting candidate to decrease its systemic toxicity.
Although the mechanism of action of Eto is similar to Dox, this drug is cell cycle dependent and phase specific, affecting mainly the S and G2 phases (19). Combination of low or IC50 concentrations of Eto caused a significant decreased of cell population in G1 phase, S phase arrest and apoptosis. However, the percent of cells undergoing apoptosis was doubled when Eto IC50 dose was combined with LiCl compared to its low dose combination (Table 2). This may suggest that use of antineoplastic drugs affecting cells in S phase in combination with LiCl would be more effective to increase tumor cell death.
The other finding of the present study was combination of LiCl with antineoplastic agents affecting microtubules organization like Vin leading to G2/M arrest that may not increase cytotoxic effect of this kind of drug (Table 3). The most interesting finding of this study is the significant increase of apoptotic cells when Eto was used in combination with LiCl.
In support of our study for usefulness of LiCl in combination therapy a very recent study showed that LiCl significantly suppressed tumor development and growth of subcutaneous xenografts derived from human prostate cancer cells (15). This study revealed that following GSK-3 inhibition, C/EBPa, a negative cell cycle regulator, was remarkably accumulated in xenograft tumors or in cultured PCa cells and knocking down C/EBPa expression abolished GSK-3ß inhibition-induced suppression of E2F1 transactivation, suggesting that C/EBPa accumulation is involved in GSK-3 inhibition-induced anti-tumor effect (15).
According to the previous reports (13,20) , it has been shown that DU145 cells are Fas (CD95) positive, Bax negative and showed abundant cytoplasmic p53 protein (12). Interestingly, LiCl induced p53 stabilization by affecting the levels of protein and not mRNA in primary endothelial cells leading to G2/M arrest of these cells. Similarly, we found increased levels of p53 which was abundant in cytoplasm and nucleus of DU145. Interestingly, this study showed significant G2/M arrest with Dox or Vin combined with LiCl (Tables 1 and 3) which may probably preceed p53 stability and /or expression. It should be noted whether the effect of LiCl on p53 affects directly or indirectly growth arrest or apoptosis, needs further investigations.
Further argument support the evidence that the antiproliferative effect of LiCl on DU145 cells could not be mediated by its interaction with AR. DU145 cells are hormone non-responsive, nevertheless it has been reported that they express AR mRNA and protein, however DHT treatment of DU145 cells did not result in transcriptional activation of AR-responsive genes (11). This may emphasize the hypothesis that anti-proliferative effect of LiCl on DU145 may not be related to AR inhibition. Of interest, previous report have demonstrated that LiCl enhances TNF-α-mediated anti-tumor activity in vitro and in vivo (31,32). In addition, it has been reported that GSK-3 inhibition by LiCl or SB216763 sensitizes dramatically PCa cell lines to TRAIL-induced apoptosis (9).
In addition, previous report showed that LiCl showed a sensitizing effect in DU145 cells that are lacking Bax expression (12), and Bcl-2 was also reported to inhibit TRAIL-mediated cell death (33). LiCl has been shown to induce p53 stabilization in endothelial cells (22) and enhanced cell death in PC-3 cells that are lacking p53 expression (12). Taken these studies it seems that Bcl-2 family or p53 pathway may not be responsible for GSK-3 inhibitor induced TRAIL sensitization. Further investigations may clarify the relationship between LiCl-induced p53 levels and TRAIL sensitization in the present study.
TRAIL may prove to be an effective antitumor agent which may enhance the effectiveness of treatment with chemotherapeutic drugs and irradiation (34). Moreover, in a study with six tumor cell line of PCa and normal epithelial cells of prostate gland it has been demonstrated that only tumor cells were killed by TRAIL and advanced tumor cells were more sensitive than early stage cells (35). LiCl significantly reduced cancer incidence compared to the controls both in clinical observation and animal studies, indicating its possible value in human cancer intervention (36-38). Moreover, LiCl as a TRAIL sensitizing agent would offer additional advantages by enhancing the cytotoxic effect of antineoplastic drugs towards hormone non-responsive PCa cell lines.
Conclusion :
This study for the first time demonstrated LiCl usefulness in combination therapy in an in vitro model, although further in vivo study using various cell cycle specific chemotherapeutic drugs in combination with LiCl and subsequent assessment of tumor growth in a mice xenograft model may warrant findings of the present study.
Acknowledgement :
We are grateful to Prof. Houri Sepehri for her support and Dr. Masoumeh Malek for helpful advises regarding statistical analysis from School of Biology, University College of Science, University of Tehran. This work was partly supported by grant from College of Science, University of Tehran.

Figure 1. Effect of LiCl A) and NaCl B) on viability of DU145 cells. Values are expressed as mean+SD from at least three independents experiments in triplicate. *: p<0.05, **: p<0.01, ***: p<0.001 compared to control
|
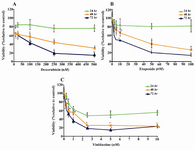
Figure 2. Dose response curves of DU145 cells after 24, 48 and 72 hr of exposure to, A) Doxorubicin, B) Etoposide and C) Vinblastine. Values are expressed as mean+SEM from at least three independents experiments in triplicate. IC50 values were calculated by using instat-3 software. a: p<0.05, b: p<0.01, c: p<0.001 compared to control
|
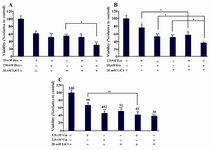
Figure 3. Effect of LiCl and antineoplastic drugs alone or in combination on DU145 cell growth. A) Doxorubicin (Dox), B) Etoposide (Eto) and C) Vinblastine (Vin), Values are expressed as meanSEM relative to control. *: p<0.05; **: p<0.01; ***: p<0.001
|

Figure 4. Cell cycle analysis of DU145 cells in the presence of LiCl or Doxorubicin alone or in combination by flow cyto-metry analysis. 230 M and 2.5 M represent IC50 and low doses, respectively. 20 mM LiCl represents IC50 dose. M1: subG1; M2: G1; M3: S; M4: G2/M phases. Figure is rep-resentative of one of three independent experiments
|

Figure 5. Cell cycle analysis of DU145 cells in the presence of LiCl or Etoposide in combination by flow cytometry analysis. 18 M and 2.5 M represent IC50 and low doses, respectively. 20 mM LiCl represents IC50 dose. M1: subG1; M2: G1; M3: S; M4: G2/M phases. Figure is representative of one of three independent experiments
|

Figure 6. Cell cycle analysis of DU145 cells in the presence of LiCl or Vinblastine in combination by flow cytometry analy-sis. 2.5 nM and 1.5 nM represent IC50 and low doses, respect-ively. 20 mM LiCl represents IC50 dose. M1: subG1; M2: G1; M3: S; M4: G2/M phases. Figure is representative of one of three independent experiments
|
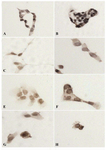
Figure 7. Subcellular localization of p53 in DU145 cells. DU145 cells were seeded in 96 wells plates overnight and then treated without or with antineoplastic and LiCl alone or in combination for 48 hr. A) control (untreated); B) LiCl
20 mM; C) Doxorubicin 230 M; D) Doxorubicin 230 M + LiCl 20 mM E) Etoposide 18 M F) Etoposide 18 M + LiCl 20 mM G) Vinblastine 5 M H) Vinblastine 5 M+LiCl
20 mM. Results showed strong nuclear p53 immunostaining in the presence of LiCl alone compared to other conditions
|
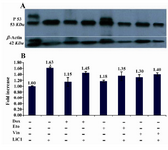
Figure 8. Increased levels of p53 protein in the presence of LiCl. DU145 cells were seeded in 6 wells plates overnight and then treated without or with antineoplastic and LiCl alone or in combination at indicated doses for 48 hr. p53 were assessed as described in the text. -actin blot served as loading control. Data are from three independent experi-ments
|
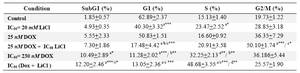
Table 1. Effect of Doxorubicin and LiCl alone or in combination on cell cycle progression of DU145 cells
a: compared to control; b: compared to 25 nM; c: compared to 20 mM; d: compared to 230 nM *: p<0.05; **: p<0.01;
***: p<0.001. Values are expressed as mean+SEM (n > 3)
|
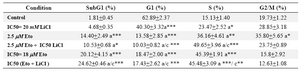
Table 2. Effect of Etoposide and LiCl alone or in combination on cell cycle progression of DU145 cells
a: compared to control; c: compared to 20 mM. *: p<0.05; **: p<0.01; ***: p<0.001. Values are expressed as mean+SEM (n > 3)
|
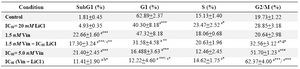
Table 3. Effect of Vinblastine and LiCl alone or in combination on cell cycle progression of DU145 cells
a: compared to control; b: compared to 2.5 nM; c: compared to 20 mM; d: compared to 1.5 nM *: p<0.05; **: p<0.01; ***: p<0.001. Values are expressed as mean+SEM (n > 3)
|
|